Open Access | Case Report
This work is licensed under a Creative
Commons Attribution-ShareAlike 4.0 International License.
Robot-assisted repair of rectovesical fistula
* Corresponding author: Alessandro Veccia
Mailing address: Department of Urology, AOUI Verona, Verona,
Piazzale Stefani 1, 37126, Italy.
Email: a.veccia88@gmail.com
Received: 08 October 2022 / Revised: 01 December 2022 / Accepted: 07 December 2022 / Published: 29 December 2022
DOI: 10.31491/UTJ.2022.12.004
Abstract
A 76-year-old man developed a rectovesical fistula after rectal resection for adenocarcinoma complicated by peritonitis with the necessity of the creation of a colostomy. The patient was undergone a rectosigmoidoscopy with the placement of clip-type Ovesco to close the fistula, but this technique resulted ineffective. A robot- assisted closure of the rectovesical fistula was performed using Xi Da Vinci System. The surgical procedure will be divided into 3 parts: longitudinal median cystotomy, excision of the fistula tract, and the closure of the anterior rectal wall and bladder with interposition of omentum, between the suture lines of the rectum and bladder, to prevent fistula recurrence. The patient removed the catheter after 4 weeks of surgery and the patient remained free of urinary leakage from the anus. In February 2021, the surgery for intestinal re-anastomosis was performed. Robotic surgery is a good approach for the treatment of rectovesical fistula when the endoscopic treatment was unresolving.
Keywords
Rectovesical fistula, prostate cancer, robotic-assisted surgery
Introduction
Enterovesical fistulas result from pathological communication between the intestine and the bladder. Fistulas
are subdivided into colovesical, rectovesical, ileocecal,
and appendicovesical, according to the intestinal segment
involved. Among these, colovesical fistulas are the most
common (95%) and occur in about 1 surgical hospitalization in 3000 with an incidence of 0.5/10,000 cases [1].
This condition is more frequent in males than a female
with a ratio of 3: 1 [2].
The leading causes of colovesical fistulas are diverticulitis
in 65% -79% of cases and Crohn’s disease. In 10-15% of
cases, the fistula is a consequence of colorectal carcinoma
or bladder cancer. Other causes are represented by surgery, such as enlargement cystoplasty, prostatectomy or
rectal resections, and chemotherapy or radiotherapy [3].
Urologically speaking, the urinary fistulas might be a
consequence of prostate surgery, mainly after radical prostatectomy. Indeed, the incidence of rectal injuries during
radical prostatectomy ranges from 1 to 11% [4].
Despite intestinal involvement, the most common symptoms of colovesical fistulas are urological, such as pneumaturia and fecaluria. Other symptoms are bladder irritability and usually a change in bowel habits. Diagnosis
could be difficult, and digital rectal examination might
lead to it. Diagnostic insights such as CT, MRI, cystourethroscopy, colonoscopy, and retrograde urethrography can
aid in diagnosis and operative planning [1]. These conditions are surgically treated in 97% of cases. The open
approach is adopted in 63.3% of cases, whereas in 35.1%
laparoscopy is the preferred one. Only 1.6% are addressed
to the robotic treatment [2].
Despite the well-known advantages of the laparoscopic
approach in terms of length of stay, earlier bowel mobility, and less morbidity, this approach could translate either
in higher complications or conversion rate because of the
local inflammation [5, 6].
Moreover, laparoscopy is a challenging technique, especially during the dissection of the fistula and the intracorporeal suture. The advent of robotics led to overcoming
the laparoscopic issues allowing fatigue-free ergonomic
maneuverability of the instruments, accuracy, and magnified three-dimensional vision. In addition, it helped to
limit the need for extensive dissection, tissue manipulation, and placement of suprapubic cystostomy, even in
cases of recurrent fistula surgery [7]. Nevertheless, this approach is still underused. This study aims to describe a
case of robot-assisted rectovesical fistula repair describing
the technique and the postoperative outcomes.
Case report
The present case is of a non-smoker 76-year-old man, with a BMI of 22. The patient underwent laparoscopic radical prostatectomy for prostate adenocarcinoma in 2009 with a follow-up negative for recurrence of the disease. In February 2020, the patient underwent laparoscopic rectal resection for adenocarcinoma (pT3b N1M0). The postoperative course was complicated by dehiscence of the anastomosis and abdominal abscess requiring exploratory laparoscopy and colostomy placement. Thereafter the patient complained of the appearance of urinary leakage from the anus. He underwent cystoscopy with the detection of a rectovesical fistula at the level of the posterior bladder wall and rectosigmoidoscopy with the placement of clip-type Ovesco. The cystography and fistulography highlighted the opacification of the rectum-sigma through the bladder due to the presence of a rectovesical fistula.
Surgical Technique
The patient underwent robot-assisted closure of the rectovesical fistula following laparoscopic rectal resection, using the Xi Da Vinci System. An endoscopic approach by placing a clip was first attempted, but it immediately failed. Therefore, a standard transperitoneal robotic approach was planned to correct the rectovesical fistula. The first step was a longitudinal median cystotomy, subject to placement of J ureteral stent as a retrieval (Figure 1).
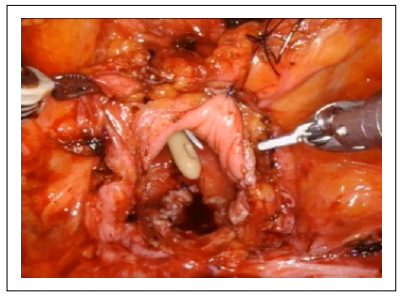
Figure 1. Cystotomy.
The opening of the fistulous tract and the dissection between the bladder and the rectum allowed the identification of both the fistula site and the clips previously applied. The fistulous tract and the surrounding inflammatory tissue were then excised (Figure 2).
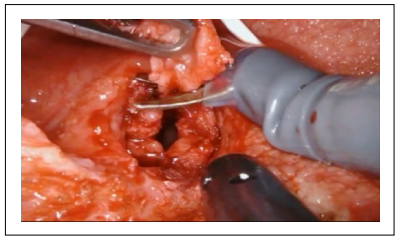
Figure 2. Excision of the fistulous tract.
Three individual layers of the urothelium, anterior rectal muscle wall, and rectal mucosa were identified and dissected free to be carried out to permit a water-tight, tension-free closure. The rectal mucosa was first closed and then the anterior rectal wall was closed with a running, 2-0 absorbable suture (Figure 3).
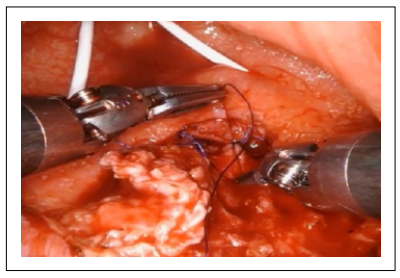
Figure 3. The anterior rectal wall was closed with a running 2-0 absorbable.
A bubble test at the end of the first layer of the rectal suture showed a water-tight closure. The interposition of the omentum was performed to prevent the fistula recurrence (Figure 4).
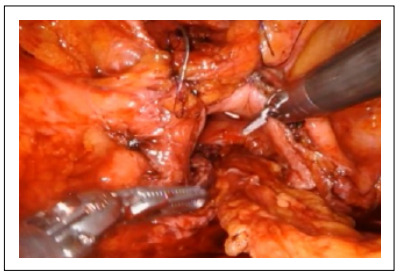
Figure 4. Interposition of the omental flap.
The urothelium was closed with a running, 3-0 absorbable suture in a similar fashion (Figure 5-6).
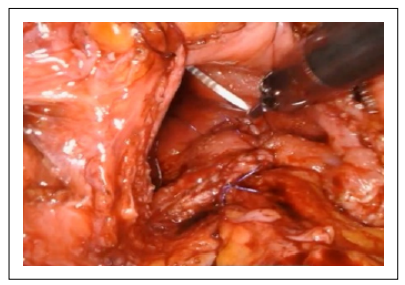
Figure 5. The urothelium was closed with a running, 3-0 absorbable suture.
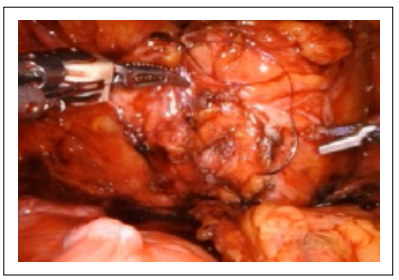
Figure 6. Cystotomy closure.
The operative time was 203 minutes. The hospital stay was 4 days. The urethral catheter was kept indwelling for 30 days, then the patient remained free of urinary leakage from the anus. In February 2021, the colostomy was closed and intestinal re-anastomosis was performed by general surgeons. At the 2-year follow-up, the patient remains free of fistula recurrence (Supplementary Video).
Discussion
As mentioned above, surgeons rarely use the robot for
correcting rectovesical fistulas: approximately, robotic
surgery represents only 1.6% of corrective interventions
compared to endoscopic and open surgery. Zizzo et al.
performed a systematic literature review, 22 included articles from 1982 to 2019, with a total of 1,365 patients.
Almost all patients with colovesical fistula underwent
open colorectal resection (97.1%) with or without partial/total cystectomy [8]. We report our experience with
a single case of robot-assisted rectovesical fistula repair
which demonstrates the procedure to be feasible and safe.
The few cases reported in the literature of robotic surgery
used to repair this type of fistula demonstrates this data.Sotelo et al. presented an iatrogenic robotic fistula repair
in a patient who underwent ischemic colitis and Hartmann’s surgery. They reported a fistula adequately treated without medium and long-term operative complications
[9]. Oderda et al. reported a similar case of our iatrogenic
rectal bladder fistula following radical prostatectomy [10].
They found the robotic correction to be safe and effective,
like in our case [7]. The few available works describing
the robotic technique to treat this condition underlined
some common denominators.
The robot Da Vinci allows an excellent view of the anatomical planes to be dissected; the magnification of the
anatomical details in this type of surgery is not to be
underestimated. If not consequent to previous surgery,
fistulas could have inflammatory or infectious etiopathogenesis. The primary conditions that determine the rectovesical fistulas, therefore, lead to an operating field that is
difficult to interpret, hampered by inflamed or fibrotic tissues, with numerous adhesions. The visual inspection of
the dissection planes of tissues that have undergone previous phenomena of inflammation and fibrosis is therefore
not trivial. In these cases, robotic surgery is advantageous
for the vision of the anatomical planes it offers the surgeon [11]. In addition, the sutures that can be performed
in small spaces, in complicated pelvic anatomy with the
movements performed by the robot allow a technical execution of excellent quality compared to other surgical
techniques. The limits of robotic surgery remain the high
costs of purchasing and managing the robot system, costs
that will, in any case, be reduced more and more in the future with the spread of these surgical devices. Even at the
post-operative checks, the fistula is corrected and repaired
without signs of recurrence.
Our technique of robotic repair of rectovesical fistula was
safe and effective.
Declarations
Authors’ contributions
All authors contributed to the conception, design of the study, and material support of the article.
Availability of data and materials
All data of the patient is stored at AOUI Verona where the operation is performed.
Financial support and sponsorship
None.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
We studied the ethical guidelines set by the Helsinki Declaration.
References
1. Scozzari G, Arezzo A, & Morino M. Enterovesical fistulas: diagnosis and management. Tech Coloproctol, 2010, 14(4): 293-300. [Crossref]
2. Cochetti G, Del Zingaro M, Boni A, Cocca D, Panciarola M, Tiezzi A, et al. Colovesical fistula: review on conservative management, surgical techniques and minimally invasive approaches. G Chir, 2018, 39(4): 195-207.
3. Golabek T, Szymanska A, Szopinski T, Bukowczan J, Furmanek M, Powroznik J, et al. Enterovesical fistulae: aetiology, imaging, and management. Gastroenterol Res Pract, 2013, 2013: 617967. [Crossref]
4. Harpster LE, Rommel FM, Sieber PR, Breslin JA, Agusta VE, Huffnagle HW, et al. The incidence and management of rectal injury associated with radical prostatectomy in a community based urology practice. J Urol, 1995, 154(4): 1435-1438.
5. Kitaguchi D, Enomoto T, Ohara Y, Owada Y, Hisakura K, Akashi Y et al. Laparoscopic surgery for diverticular colovesical fistula: single-center experience of 11 cases. BMC Res Notes, 2020, 13(1): 177. [Crossref]
6. Martinolich J, Croasdale DR, Bhakta AS, Ata A, Chismark AD, Valerian BT, et al. Laparoscopic Surgery for Diverticular Fistulas: Outcomes of 111 Consecutive Cases at a Single Institution. J Gastrointest Surg, 2019, 23(5): 1015- 1021. [Crossref]
7. Delto, J. C., Hemal, A. K., & Bhandari, A. Robotic repair of urinary fistulae. In Robotics in Genitourinary Surgery, Second Edition, 2018. [Crossref]
8. Zizzo M, Tumiati D, Bassi MC, Zanelli M, Sanguedolce F, Porpiglia F, et al. Management of colovesical fistula: a systematic review. Minerva Urol Nephrol, 2022, 74(4): 400-408. [Crossref]
9. Sotelo R, Medina LG, Husain FZ, Khazaeli M, Nikkhou K, Cacciamani GE, et al. Robotic-assisted laparoscopic repair of rectovesical fistula after Hartmann’s reversal procedure. J Robot Surg, 2019, 13(2): 339-343. [Crossref]
10. Oderda M, Bonet X, Campobasso D, & Gaston R. Robotic rectovesical fistula repair: a successful approach. J Laparoendosc Adv Surg Tech A, 2014, 24(8): 567-570. [Crossref]
11. Antonelli A, Veccia A, Morena T, Furlan M, Peroni A, & Simeone C. Robot-assisted vesico-vaginal fistula repair: technical nuances. Int Braz J Urol, 2021, 47(2): 684-685. [Crossref]