Open Access | RESEARCH
This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.
Low- vs high-load power training on muscle quality, functional performance, and multi-omic profiles in frail older adults: protocol for the FrailPow randomized controlled trial
* Corresponding author: Xabier Río
Mailing address: Faculty of Education and Sport. University of Deusto. Avda. Universidades 24. 48007. Bilbao, Spain.
Email: Faculty of Education and Sport. University of Deusto. Avda. Universidades 24. 48007. Bilbao, Spain.
Received: 08 December 2025 / Revised: 06 January 2026 / Accepted: 27 January 2026 / Published: 31 March 2026
DOI: 10.31491/APT.2026.03.206
Abstract
Background:Age-related frailty represents a growing global public health challenge, as it is associated with increased risk of falls, disability, and mortality, directly impacting functional decline and quality of life in older adults. Although resistance training is widely recommended as a therapeutic strategy, evidence on the differential effects of power training, particularly low- versus high-load modalities, compared with conventio
Methods:This study will be a three-arm, multicenter, randomized controlled trial. A total of 207 older adults (≥ 70 years) meeting frailty criteria will be recruited from the Osakidetza healthcare network (Basque Country, Spain). Participants will be randomly assigned to: (1) low-load power training, (2) high-load power training, or (3) traditional resistance training. Each intervention will consist of a 12-week personalized exercise program.
Results: This manuscript reports the protocol of the FrailPow randomized controlled trial; therefore, no outcome data are available at the time of submission. The study will quantify longitudinal changes across predefined assessment time points in muscle-related outcomes, functional performance, body composition, and multi-omics biomarkers.
Conclusion:FrailPow will generate robust evidence on the efficacy and safety of power training in frail older adults, distinguishing the effects of load intensity. Additionally, the study will explore the potential of multiomics biomarkers and advanced muscle quality metrics as tools to monitor and personalize interventions. Findings will be transferable to clinical and community settings, supporting precision exercise strategies within the Basque public health system to promote healthy aging.
Trial registration: Registered on the Open Science Framework platform: https://osf.io/yrz4h/overview?view_only=802a2e066c1a4c85bc43d0d4ceb3b494.
Keywords
Frailty, muscle quality, power training, functional performance, multi-omics, gut microbiota
Introduction
Population ageing is one of the major public health challenges of the 21st century. The rapid growth of adults aged 60 years and older is expected to increase the burden of chronic disease, disability, and dependence. This demographic shift emphasizes the need to move from a diseasecentered model of care toward approaches that preserve functional capacity and autonomy in later life [1, 2]. The distinction between lifespan and healthspan highlights that additional years of life should be lived in good health [3]. Recent estimates from the Global Burden of Disease project indicate that in many European countries, gains in healthy life expectancy are stagnating or even declining, partly due to preventable causes and modifiable risk factors [4]. These trends underline the importance of prioritizing the prevention of frailty and functional decline as central goals for healthy ageing strategies and clinical practice [5].
Frailty is a common geriatric syndrome characterized by reduced physiological reserve and increased vulnerability to adverse outcomes such as disability, hospitalization, and mortality [6]. Two operational models are widely used. The physical frailty phenotype described by Fried defines frailty through five criteria: low grip strength, slow gait speed, exhaustion, unintentional weight loss, and low physical activity [7]. The deficit-accumulation model conceptualizes frailty as a multidimensional condition involving medical, functional, and cognitive impairments [8, 9]. Sarcopenia, defined by the European Working Group on Sarcopenia in Older People 2 (EWGSOP2) as a progressive loss of muscle strength and mass, is considered one of the core biological substrates of physical frailty and contributes substantially to mobility limitations and dependence in older adults [10, 11]. Although frailty and sarcopenia overlap, they are not equivalent. Frailty reflects broader systemic and functional decline, whereas sarcopenia refers specifically to musculoskeletal deterioration [12, 13].
Loss of muscle strength and power, commonly referred to as dynapenia, appears earlier and progresses faster with ageing than reductions in muscle mass, underscoring the central role of neuromuscular impairments in functional decline [14, 15]. Muscle quality, defined as the ability of muscle tissue to generate force or power relative to its size and composition, has therefore emerged as a more sensitive indicator of mobility, disability risk, and functional independence than muscle mass alone [16, 17]. Structural and functional alterations, including fiber-type atrophy, fatty infiltration, and reduced contractile efficiency, contribute to this deterioration and highlight the need for interventions that target neuromuscular performance rather than focusing solely on increasing muscle quantity [18-20].
Traditional resistance training (RT) is typically performed with moderate-to-high loads lifted at controlled, slow velocities to maximize strength through progressive mechanical overload [21, 22]. In contrast, power training (PT) focuses on the rapid production of force using submaximal loads moved at high velocity, directly targeting neuromuscular attributes that decline earliest with ageing, including rate of force development and type II fiber function [23, 24]. Evidence shows that PT produces greater improvements in movement speed, balance recovery, and functional performance in older adults than conventional strength-focused traditional RT [25, 26]. However, most trials do not distinguish between low-load and high-load PT, leaving uncertainty about their relative efficacy for improving muscle quality and mobility in frail populations.
Despite growing interest in power-oriented exercise for older adults, important gaps in the evidence remain. Many randomized trials include small samples, brief intervention periods, and heterogeneous protocols that do not distinguish between low- and high-load PT, limiting conclusions about their relative effectiveness [25-27]. In addition, few studies assess functional, clinical, and biological outcomes together, restricting understanding of the mechanisms underlying exercise responsiveness in frail older adults [28, 29]. These gaps underline the need for rigorously designed interventions that can clarify how different PT modalities influence muscle quality and functional trajectories in populations at elevated risk of disability.
The present randomized controlled trial (RCT) is designed to address these gaps by directly comparing low-load PT, high-load PT, and traditional RT on muscle quality in frail older adults. By integrating functional performance, body composition, and biological markers within a unified assessment framework, the study aims to clarify the adaptive responses to different neuromuscular stimuli and to guide more individualized exercise prescriptions for populations at high risk of functional decline.
Methods
Study design, setting, and sample size
The FrailPow study is a multicenter, parallel-group RCT designed to compare low-load PT, high-load PT, and traditional RT on muscle quality in frail older adults. The trial includes three phases: (1) baseline assessment before randomization, (2) a 12-week supervised intervention, and (3) follow-up assessments at weeks 24 and 36. The overall study flow is illustrated in Figure 1.
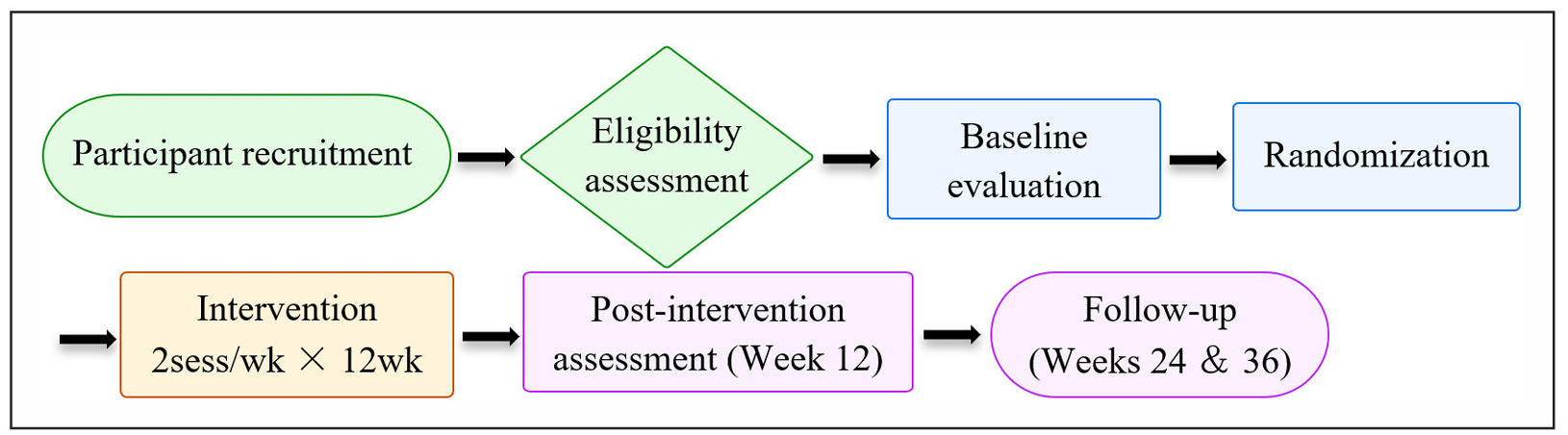
Figure 1. General flow diagram of the FrailPow RCT according to CONSORT 2025.
The study will be conducted within the public primary care network of the Basque Health System (Osakidetza), in collaboration with the University of Deusto and the Biobizkaia Health Research Institute (BHRI). Potential participants will be identified and screened in primary care centers of the Bilbao-Basurto Integrated Healthcare Organization (HIO). Functional assessments and training sessions will be performed at the Faculty of Education and Sport of the University of Deusto, while the processing and storage of biological samples will be conducted at BHRI.
The target sample size is 207 participants (69 per arm). This provides 80% power to detect a minimum clinically important difference of 0.6 units in the muscle quality index (MQI) with a two-sided alpha of 0.05, assuming a 20% dropout rate during follow-up. The MQI is defined as an indirect functional index derived from Sit-to-Stand test (STS). Sample size calculations were performed in G*Power using a moderate effect size (f = 0.25) for three intervention groups, consistent with effect sizes typically observed in high-velocity resistance training interventions in older adults [25, 30]. The operational timeline of study phases and assessments is presented in Table 1.
Table 1.
perational timeline of the FrailPow RCT
| Study phase / evaluation | Prerecruitment | Baseline (week 0) | Intervention¹ (week 12) | Follow-up (week 24) | Follow-up 2 (week 36) |
|---|---|---|---|---|---|
| Recruitment and informed consent | √ | – | – | – | – |
| Telephone screening and clinical eligibility | √ | – | – | – | – |
| Random allocation | – | √ | – | – | – |
| Sociodemographic and clinical measures | – | √ | – | – | – |
| Anthropometric and body composition assessment | – | √ | √ | √ | √ |
| Muscle quality assessment | – | √ | √ | √ | √ |
| Functional assessment | – | √ | √ | √ | √ |
| Quality of life questionnaire | – | √ | √ | √ | √ |
| Blood sampling | – | √ | √ | √ | √ |
| Fecal sample collection | – | √ | √ | √ | √ |
| Supervised intervention | – | – | √ (weeks 1-12) | – | – |
| Monitoring of adherence and safety | – | √ | √ (continuous) | √ | √ |
| Adverse event recording | – | √ | √ (continuous) | √ | √ |
| Data analysis and dropout tracking | – | √ | √ (continuous) | √ | √ |
| Final statistical analysis and multivariate integration | – | – | – | – | √ |
| Dissemination of results | – | – | – | – | √ |
Note: 1Intervention sessions occur twice per week during weeks 1–12.
Randomization and allocation concealment
Participants who meet all eligibility criteria will be randomly assigned to one of the three intervention groups using a computer-generated permuted-block sequence stratified by sex and frailty level. The randomization list will be prepared by an independent researcher with no involvement in recruitment, assessments, or intervention delivery. Allocation concealment will be ensured through a centralized electronic system, accessible only to the data manager, and group assignment will remain concealed until participant enrollment. Outcome assessors will be blinded to group allocation throughout all evaluation phases.
Participants, eligibility criteria, and recruitment
The study will recruit community-dwelling older adults aged 70 years or older who are registered within the Osakidetza primary care network. Potential candidates will be identified by family physicians, nurses, and physiotherapists during routine geriatric assessments and active ageing programs within the HIO.
Recruitment will follow a two-step screening procedure. First, an initial telephone or in-person interview will be conducted to exclude individuals with medical contraindications. Candidates who remain eligible will then undergo a comprehensive baseline evaluation to determine frailty status and complete cognitive screening, body composition analysis, muscle quality assessments, and functional performance tests. The overall screening and enrollment process is illustrated in Figure 2.
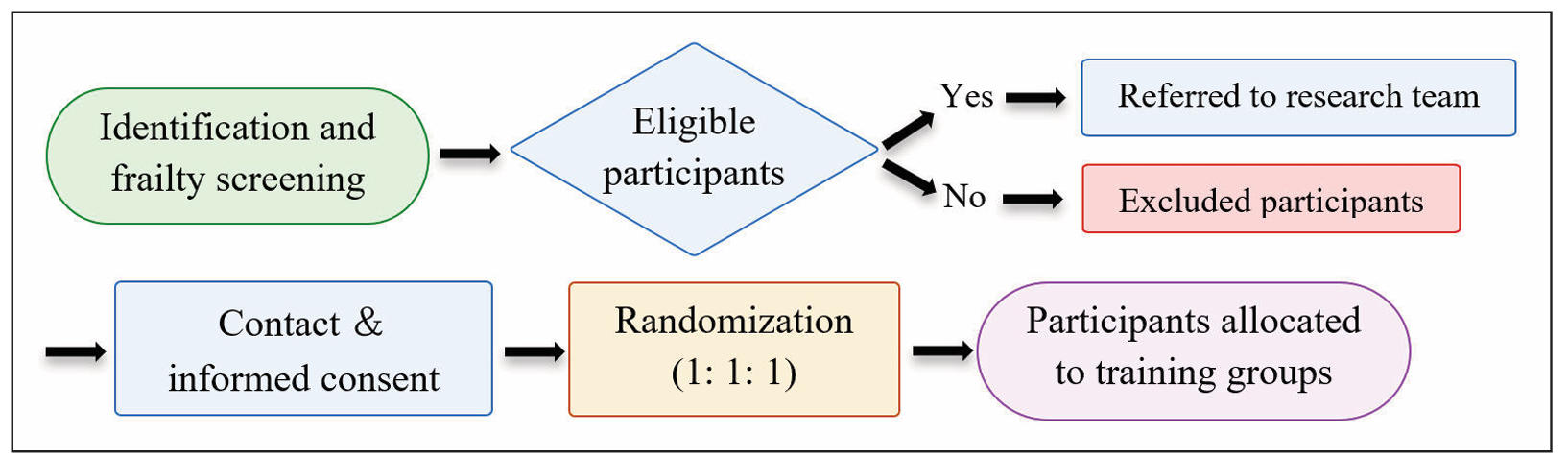
Figure 2. Flow diagram of participant screening, randomization, and follow-up for the FrailPow trial, structured according to CONSORT 2025 guidelines.
A detailed summary of inclusion, exclusion, and termination criteria is provided in Table 2, which serves as the operational reference for all definitions and thresholds usedin this trial.
Table 2.
Inclusion and exclusion criteria for the FrailPow RCT
| Type of criterion | Specific criterion |
|---|---|
| Inclusion | • Age ≥ 70 years |
| • Barthel index ≥ 90 | |
| • SPPB < 10; TUG > 12; GS < 0.8 m/s | |
| • Diagnosis of frailty: meeting ≥ 3; modified Fried criteria or FRAIL Index ≥ 3 points | |
| • Adequate physical and cognitive capacity to perform supervised exercise | |
| Exclusion | • Severe neuromuscular disorders or unstable cardiac conditions |
| • Severe cognitive impairment (MMSE < 24) | |
| • Active oncological processes or treatment | |
| • Absolute medical contraindications for exercise | |
| • Participation in supervised exercise programs within the last 6 months | |
| • Current institutionalization or dependence on home-based nursing care | |
| • Terminal condition or receipt of palliative care | |
| • Absence of caregiver or support for follow-up requirements | |
| Termination | • Withdrawal of consent at any time |
| • Medical complications or adverse events requiring discontinuation | |
| • Non-adherence compromising participant safety |
Intervention
Baseline assessments will be performed 2–3 weeks before randomization and will include body composition using multifrequency bioelectrical impedance analysis (BIA) [31, 32]; muscle quality assessments through tensiomyography (TMG) [33] and phase angle (PhA) derived from BIA [34, 35]; and functional tests widely validated in geriatric research, including the Short Physical Performance Battery (SPPB) [36], Timed Up and Go (TUG) [37], gait speed (GS) [38], STS [39], and handgrip strength (HGS) [40]. All evaluations will be distributed across two separate days to minimize fatigue and ensure consistent measurement.
To reduce fatigue and standardize conditions, assessments will be split into two evaluation sessions. On Day 1, participants will complete functional related tests. On Day 2, body composition assessment by multifrequency BIA and muscle contractile assessment by TMG will be conducted. Standardized rest periods will be provided between tests and trials, and additional recovery time will be allowed when needed to ensure safe and valid performance in this frail population.
After baseline assessments, all participants will complete a two-week familiarization period (two sessions per week). This phase ensures adequate technical execution, reduces injury risk, and standardizes movement patterns before exposure to the assigned training stimulus. Following familiarization, participants will be randomly allocated to one of the three intervention groups.
The intervention will consist of 12 weeks of supervised training, with two non-consecutive sessions per week (≈ 60 minutes), ensuring at least one rest day between sessions. Each session will include a warm-up, a main training phase, and a cool-down. Training will take place at the University of Deusto under the supervision of qualified exercise professionals.
All groups will perform the same exercise selection: smith machine press, smith machine horizontal pull (assisted half pull-up variation), squat, and supported lunge. The intervention arms will differ only in load, movement velocity, repetition range, and the intended neuromuscular stimulus.
Initial training loads will be estimated using a linear encoder system, allowing adjustment of relative intensity (% 1RM) based on real-time bar velocity and ensuring alignment between prescribed and achieved intensity [41, 42]. Subjective internal load will be monitored using the Borg 0–10 Rating of Perceived Exertion (RPE) scale. Load progression will follow a controlled linear pattern, with planned increases of 5–10% every three weeks, and will be determined through session-by-session monitoring of execution quality and safety in combination with objective (bar velocity) and subjective (RPE) indicators. When participants complete the prescribed sets and repetitions with consistent technique and without safety concerns, loads will be increased within the planned range; otherwise, the load will be maintained until progression criteria are met. Training fidelity, including load, repetitions, sets, rest intervals, RPE, and any adverse or technical incidents, will be recorded digitally to ensure adherence to the protocol. Table 3 summarizes the training parameters for the three intervention groups across the 12-week program.
Table 3.
Training parameters and load progression of the FrailPow intervention groups (low-load power, high-load power, and traditional resistance).
| Group | Weeks 0-3 | Weeks 3-6 | Weeks 6-9 | Weeks 9-12 |
|---|---|---|---|---|
| Low-load PT1 | 2–3 sets × 6–8 reps @ 20% 1RM Rest: 120 s |
2–3 sets × 6–8 reps @ 30% 1RM Rest: 120 s |
2–3 sets × 6–8 reps @ 40% 1RM Rest: 120 s |
2–3 sets × 6–8 reps @ 40% 1RM Rest: 120 s |
| High-load PT2 | 2–3 sets × 4–6 reps @ 60% 1RM Rest: 120 s |
2–3 sets × 4–6 reps @ 70% 1RM Rest: 120 s |
2–3 sets × 4–6 reps @ 80% 1RM Rest: 120 s |
2–3 sets × 4–8 reps @ 80% 1RM Rest: 120 s |
| Traditional RT3 | 1–2 sets × 10–15 reps @ 70–80% 1RM Rest: 120 s |
2–3 sets × 6–12 reps @ 75–80% 1RM Rest: 120 s |
2–3 sets × 6–10 reps @ 80–85% 1RM Rest: 120 s |
2–3 sets × 6–10 reps @ 80–85% 1RM Rest: 120 s |
Note: 1Performed at maximal intended concentric velocity to prioritize movement explosiveness and neuromuscular activation with low external load. 2Performed at maximal intended concentric velocity to emphasize power output and type II fiber recruitment using moderate-to-high loads. 3 Active control group performing exercises with controlled tempo (≈ 2 s concentric / 2 s eccentric), focusing on strength development and functional hypertrophy.
Safety and monitoring
To ensure participant safety, all training sessions will be supervised by qualified personnel, and equipment (guided machines, benches, pulleys, encoders, dynamometry devices, and force platforms) will undergo regular maintenance and calibration according to safety standards for older adults. Before each session, participants will complete a brief structured screening covering health status, fatigue, and musculoskeletal symptoms.
All adverse events will be documented in the electronic case report form (eCRF) and reviewed by the study coordinator. Serious adverse events will be reported to the Research Ethics Committee on Medicines of Euskadi (CEIm-Euskadi) in accordance with regulatory requirements, and participants will be withdrawn or referred for medical evaluation when clinically indicated. Adherence will be calculated as the percentage of completed sessions, with ≥ 80% considered satisfactory.
Assessment procedures and outcome measures
As described in the Intervention section, outcome assessments will be distributed across two separate evaluation days to minimize fatigue and ensure safe and consistent measurement in this frail population.
All outcomes and assessment procedures described in this protocol will be systematically collected at the prespecified assessment time points (baseline, post-intervention, and follow-up assessments), including functional performance tests, body composition and muscle mass estimation (BIA-derived measures), and multi-omics biomarkers (blood and stool samples). Outcome assessments will be conducted by trained assessors blinded to group allocation; intervention delivery will be supervised by qualified exercise professionals.
Muscle quality will be evaluated using both direct and indirect approaches. Direct assessments will include TMG of the vastus lateralis, vastus medialis, and gastrocnemius muscles [33], and through PhA derived from BIA [34, 35]. In addition, an indirect MQI will be computed using the STS test-derived equation: MQI = [(L − 0.5) × body mass × g × 5] / TSTS, where TSTS is the time (s) to complete the 5-repetition STS test, body mass is in kg, g is gravitational acceleration (9.81 m/s2), and L denotes chair height (m) [39, 43]. No additional weighting is applied beyond this equation.
Functional performance will be assessed using validated geriatric measures, including the SPPB [36], TUG test [37], GS [38], STS test [39], and HGS [40]. These tests are reliable predictors of frailty, falls, and mobility limitations and are widely used in exercise trials involving older adults.
Body composition will be evaluated using multifrequency BIA, complemented by anthropometric measures following International Society for the Advancement of Kinanthropometry (ISAK) protocols, to estimate skeletal muscle mass, fat mass, and derived indices such as the Appendicular Lean Mass Index (ALMI) [31, 32, 44].
Biomarker analysis will include genomic, transcriptomic, and proteomic markers related to inflammation, cellular senescence, metabolic regulation, and frailty-associated pathways, processed under validated laboratory protocols at the BHRI [45, 46].
Gut microbiota composition will be characterized through 16S rRNA sequencing and analyzed using measures of α-diversity, β-diversity, and taxonomic abundance from phylum to species level [47].
Health-related quality of life will be assessed using the EuroQol EQ-5D-5L questionnaire [48].
Baseline sociodemographic and clinical descriptors, including age, sex, educational level, living situation, comorbidities, medication use, physical activity, recent hospitalizations, and nutritional status, will be collected to characterize the sample and verify baseline comparability between groups.
Statistical analysis plan
All analyses will be performed in R. Data management will follow good-practice procedures, including database verification and reproducible workflows. Variable distributions will be checked with descriptive statistics and graphical methods to guide the choice between parametric and non-parametric tests.
Baseline characteristics will be summarized using means and standard deviations, medians and interquartile ranges, or frequencies and percentages. Group comparability at baseline will be examined using one-way ANOVA, Kruskal–Wallis, or Chi-square/Fisher’s exact tests, depending on variable type.
Changes in the muscle quality index (MQI) across time (baseline, week 12, week 24, week 36) and between intervention groups will be examined using repeated-measures ANOVA or their non-parametric equivalents, including group, time, and group × time interaction terms. When applicable, adjusted post-hoc comparisons and effect sizes (η2, Cohen’s d) will be reported.
The same analytical approach will be applied to functional performance, body composition, and biomarker variables.
Exploratory machine-learning procedures (e.g., Random Forest, XGBoost, clustering) will be used to identify response patterns and classify adaptation profiles, interpreted as hypothesis-generating.
Missing data patterns will be evaluated to determine if values are Missing Completely at Random or Missing at Random. Depending on the pattern and extent, completecase analysis or simple imputation will be applied. Outliers will be reviewed individually, and sensitivity analyses will be conducted when needed.
Adherence will be expressed as the percentage of completed sessions, and adverse events will be summarized descriptively across groups. Figure 3 illustrates the overall analytical workflow.
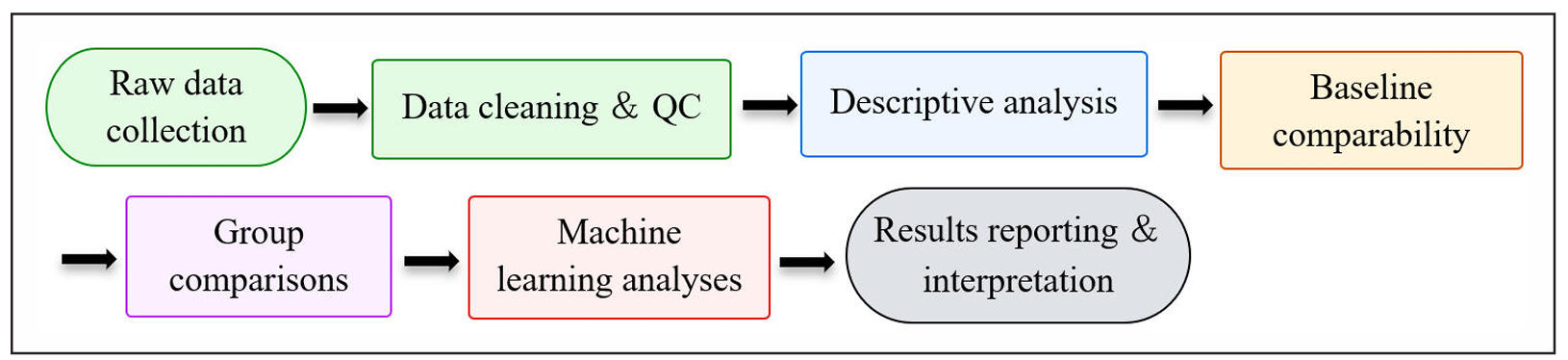
Figure 3. Statistical analysis workflow for the FrailPow RCT, developed according to CONSORT 2025 and SPIRIT 2025 recommendations.
Data will be pseudonymized and stored on secure servers with restricted access, following the General Data Protection Regulation and Good Clinical Practice principles. REDCap will be used as the central platform for data capture and quality control. Biological samples will be processed and stored according to national biobanking regulations. Serious adverse events will be reported to the ethics committee as required.
Expected outcomes
This trial is designed to test whether PT interventions induce greater improvements in muscle quality than traditional RT in frail older adults. Specifically, the study will examine changes in muscle contractile properties assessed by TMG and in PhA derived from BIA as indicators of neuromuscular function and cellular integrity. Differences between high-load and low-load PT modalities will be explored, with particular attention to neuromuscular responsiveness, tolerance, and feasibility in this population.
The study will also assess changes in functional performance across mobility- and strength-related tests and determine the extent to which these adaptations are maintained at follow-up assessments. Secondary analyses will examine accompanying changes in body composition parameters.
Exploratory analyses will investigate whether PT interventions are associated with modulation of circulating biomarkers related to inflammation, metabolism, and aging, as well as changes in gut microbiota composition and diversity. These analyses aim to contribute to a more comprehensive characterization of the multidimensional response to PT interventions in frail populations.
In addition, adherence rates and adverse events will be monitored to evaluate the feasibility and safety of implementing PT-based protocols in frail older adult populations.
Discussions
FrailPow addresses a critical gap in exercise geroscience by directly comparing low- and high-load PT with traditional RT in frail older adults. Although PT is increasingly recognized for its relevance in mobility, balance recovery, and preservation of independence in aging populations [24], few trials have differentiated load-specific power modalities or examined their specific effects on muscle quality. Current evidence suggests that neuromuscular deterioration, rather than loss of muscle mass alone, contributes substantially to the functional decline observed in frailty, underscoring the need for interventions that target rate of force development and type II fiber function [14, 15].
A key contribution of FrailPow is the multidimensional operationalization of muscle quality. By integrating TMGderived contractile properties [33], phase angle as an index of cellular integrity [34, 35], an indirect functional MQI (STS-derived) [39, 43], the protocol aligns with contemporary recommendations emphasizing combined assessment of muscle mass, quality, and function as core determinants of mobility and frailty status [11,18]. This framework moves beyond strength-only or mass-focused paradigms and enables a more comprehensive characterization of neuromuscular aging. Additionally, the selection of functional assessments is supported by reference values previously validated in the Basque Health System, including SPPB [49], GS [50], and HGS [51], reinforcing the contextual relevance of the outcomes.
The inclusion of multi-omics biomarkers represents another important methodological advance. Through genomic, transcriptomic, proteomic, and inflammatory profiling, the protocol aligns with FRAILOMIC and other European initiatives aimed at identifying biological signatures associated with frailty trajectories and responsiveness to intervention [45, 46, 52]. The incorporation of gut microbiota analysis further strengthens this integrative approach, supported by emerging evidence linking microbial ecosystems with inflammation, metabolic regulation, and muscle function in older adults [53, 54]. Together, these components position FrailPow as a precision-exercise trial capable of exploring connections between clinical adaptations and underlying biological mechanisms.
Methodologically, the protocol shows several strengths. The randomized, multicenter design with stratified allocation and blinded assessment enhances internal validity. Maintaining identical training frequency, volume, and exercise selection across intervention arms while manipulating only load and movement velocity ensures rigorous control of neuromuscular stimuli. Conducting the trial within Osakidetza increases real-world applicability and scalability within a public healthcare system. The use of a centralized digital platform for data capture supports traceability, consistency, and quality control across study sites.
From an implementation perspective, FrailPow was designed to accommodate the operational constraints commonly encountered in older adult care centers. The use of two supervised, non-consecutive training sessions per week, conservative load progression criteria, and assessment schedules distributed across separate days aims to minimize fatigue while maintaining safety and feasibility in frail populations. These design features may facilitate integration of PT approaches into routine care-center exercise programs, should effectiveness be confirmed.
Several potential limitations should be acknowledged. Heterogeneity in comorbidity burden, medication use, and baseline functional capacity may influence individual responsiveness despite stratified randomization. Participant blinding is not feasible in exercise interventions, although assessor blinding helps mitigate detection bias. The 12- week intervention and 36-week follow-up may not fully capture long-term sustainability of neuromuscular or biological adaptations. Adherence challenges may arise due to intercurrent illness or mobility constraints common in frail populations. Finally, although the integration within the Basque Health System enhances ecological validity, replication in more diverse healthcare and cultural contexts will be needed to evaluate generalizability.
Future research should examine longer-term adaptation trajectories, validate biological response profiles in larger cohorts, and explore adaptive or AI-supported exercise models. Such developments may contribute to more refined precision-exercise strategies aimed at mitigating frailty and supporting healthier aging.
Conclusions
The FrailPow trial proposes an innovative and comprehensive framework to advance precision exercise interventions for frail older adults. By integrating muscle quality assessment, multimodal biomarker profiling, and gut microbiota analysis within a randomized and longitudinal design, the study is expected to generate mechanistic and clinically relevant insights into the comparative effects of low- and high-load PT versus traditional RT.
Beyond evaluating efficacy, the trial is structured to evaluate feasibility, adherence, and safety within a public healthcare context, supporting real-world implementation of structured exercise programs for frailty prevention and management. The combination of functional, contractile, and molecular indicators may help identify distinct biological response profiles and contribute to the development of more personalized intervention strategies.
Ultimately, FrailPow aims to strengthen the translational bridge between geroscience and clinical practice, fostering evidence-informed approaches to healthy aging and contributing to the reduction of functional decline and healthcare burden associated with frailty in older adults.
Declarations
Acknowledgments
The authors express their sincere gratitude to Verónica Tíscar, Research and Innovation Coordinator at the Bilbao-Basurto HIO within IIS Biocruces Bizkaia, for her support, availability, and facilitation throughout the development of this project.
Author contributions
Xabier Río (PI at the University of Deusto) contributed to conceptualization, methodology, investigation, writing – original draft, writing – review & editing, visualization, supervision, project administration, and funding acquisition. Iker Muñoz contributed in methodology, investigation, data curation, writing – review & editing, and supervision. Aitor Santisteban-Leguina contributed to methodology, investigation, and writing – review & editing. Xabier Mateos-Marañón contributed in investigation, writing – original draft, and writing – review & editing. Arkaitz Larrinaga-Undabarrena contributed to investigation and writing – review & editing. José Ramón Sánchez-Isla contributed in investigation and writing – review & editing. Iker Sáez contributed to investigation and writing – review & editing. Oihana Hernández Ortiz de Urbina, Amaia Fernandez Alonso, Ana María López Rodríguez, Celia Aragón Ferrao, and Irene Gómez Soto each contributed to investigation and writing – review & editing. Finally, Iñaki Martín-Lesende (PI at Bilbao-Basurto HIO) contributed in investigation, writing – review & editing, supervision, and project administration.
Availability of data and materials
Data will be available from the corresponding authors upon reasonable request. After study completion, anonymized datasets and methodological materials will be deposited in the OSF repository.
Financial support and sponsorship
This study is supported by the Department of Health of the Basque Government (project No. 2025333038).
Conflicts of interest
The authors declare no conflicts of interest.
Ethical approval and informed consent
The study protocol was approved by the CEIm Euskadi, approval number PI2025187. Written informed consent will be obtained from all participants, in accordance with the Declaration of Helsinki.
References
1. Organization WH (2025). Ageing and health from https://www.who.int/news-room/fact-sheets/detail/ ageing-and-health (accessed 06.12.2025).
2. Beard J, Officer A, de Carvalho I, Sadana R, Pot A, Michel J, et al. The world report on ageing and health: a policy framework for healthy ageing. Lancet, 2016, 387(10033): 2145-2154. [Crossref]
3. Crimmins E. Lifespan and healthspan: past, present, and promise. Gerontologist, 2015, 55(6): 901-911. [Crossref]
4. Changing life expectancy in European countries 19902021: a subanalysis of causes and risk factors from the Global Burden of Disease Study 2021. Lancet Public Health, 2025, 10(3): e172-e188. [Crossref]
5. Hoogendijk E, Afilalo J, Ensrud K, Kowal P, Onder G, & Fried L. Frailty: implications for clinical practice and public health. Lancet, 2019, 394(10206): 1365-1375. [Crossref]
6. Cesari M, Prince M, Thiyagarajan J, De Carvalho I, Bernabei R, Chan P, et al. Frailty: an emerging public health Xabier Mateos-Marañón priority. J Am Med Dir Assoc, 2016, 17(3): 188-192. [Crossref]
7. Fried L, Tangen C, Walston J, Newman A, Hirsch C, Gottdiener J, et al. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci, 2001, 56(3): M146-156. [Crossref]
8. Bergman H, Ferrucci L, Guralnik J, Hogan D, Hummel S, Karunananthan S, et al. Frailty: an emerging research and clinical paradigm—issues and controversies. J Gerontol A Biol Sci Med Sci, 2007, 62(7): 731-737. [Crossref]
9. Chen X, Mao G, & Leng S. Frailty syndrome: an overview. Clin Interv Aging, 2014, 9: 433-441. [Crossref]
10. Cruz-Jentoft A, Bahat G, Bauer J, Boirie Y, Bruyère O, Cederholm T, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing, 2019, 48(1): 16-31. [Crossref]
11. Larsson L, Degens H, Li M, Salviati L, Lee Y, Thompson W, et al. Sarcopenia: aging-related loss of muscle mass and function. Physiol Rev, 2019, 99(1): 427-511. [Crossref]
12. Landi F, Calvani R, Cesari M, Tosato M, Martone A, Bernabei R, et al. Sarcopenia as the biological substrate of physical frailty. Clin Geriatr Med, 2015, 31(3): 367-374. [Crossref]
13. Reijnierse E, Trappenburg M, Blauw G, Verlaan S, de van der Schueren M, Meskers C, et al. Common ground? The concordance of sarcopenia and frailty definitions. J Am Med Dir Assoc, 2016, 17(4): 371.e377-312. [Crossref]
14. Clark B, & Manini T. Sarcopenia =/= dynapenia. J Gerontol A Biol Sci Med Sci, 2008, 63(8): 829-834. [Crossref]
15. Goodpaster B, Park S, Harris T, Kritchevsky S, Nevitt M, Schwartz A, et al. The loss of skeletal muscle strength, mass, and quality in older adults: the health, aging and body composition study. J Gerontol A Biol Sci Med Sci, 2006, 61(10): 1059-1064. [Crossref]
16. Fragala M, Kenny A, & Kuchel G. Muscle quality in aging: a multi-dimensional approach to muscle functioning with applications for treatment. Sports Med, 2015, 45(5): 641-658. [Crossref]
17. McGregor R, Cameron-Smith D, & Poppitt S. It is not just muscle mass: a review of muscle quality, composition and metabolism during ageing as determinants of muscle function and mobility in later life. Longev Healthspan, 2014, 3(1): 9-21. [Crossref]
18. Reinders I, Murphy R, Koster A, Brouwer I, Visser M, Garcia M, et al. Muscle quality and muscle fat infiltration in relation to incident mobility disability and gait speed decline: the age, gene/environment susceptibility-reykjavik study. J Gerontol A Biol Sci Med Sci, 2015, 70(8): 1030-1036. [Crossref]
19. Visser M, Goodpaster B, Kritchevsky S, Newman A, Nevitt M, Rubin S, et al. Muscle mass, muscle strength, and muscle fat infiltration as predictors of incident mobility limitations in well-functioning older persons. J Gerontol A Biol Sci Med Sci, 2005, 60(3): 324-333. [Crossref]
20. Radaelli R, Taaffe D, Newton R, Galvão D, & Lopez P. Exercise effects on muscle quality in older adults: a systematic review and meta-analysis. Sci Rep, 2021, 11(1): 21085. [Crossref]
21. Lopez P, Pinto R, Radaelli R, Rech A, Grazioli R, Izquierdo M, et al. Benefits of resistance training in physically frail elderly: a systematic review. Aging Clin Exp Res, 2018, 30(8): 889-899. [Crossref]
22. Fiatarone M, Marks E, Ryan N, Meredith C, Lipsitz L, & Evans W. High-intensity strength training in nonagenarians. effects on skeletal muscle. JAMA, 1990, 263(22): 3029-3034.
23. Izquierdo M, & Fiatarone Singh M. Promoting resilience in the face of ageing and disease: the central role of exercise and physical activity. Ageing Res Rev, 2023, 88: 101940. [Crossref]
24. Izquierdo M, & Cadore E. Power to prolong independence and healthy ageing in older adults. Br J Sports Med, 2024, 58(10): 524-526. [Crossref]
25. Balachandran A, Steele J, Angielczyk D, Belio M, Schoenfeld B, Quiles N, et al. Comparison of power training vs. traditional strength training on physical function in older adults: a systematic review and meta-analysis. JAMA Netw Open, 2022, 5(5): e2211623. [Crossref]
26. El Hadouchi M, Kiers H, de Vries R, Veenhof C, & van Dieën J. Effectiveness of power training compared to strength training in older adults: a systematic review and meta-analysis. Eur Rev Aging Phys Act, 2022, 19(1): 18-32. [Crossref]
27. Bandeira-Guimarães M, Blanco-Rambo E, Vieira A, Sáez de Asteasu M, Pinto R, Izquierdo M, et al. Chronic effects of different intensities of power training on neuromuscular parameters in older people: a systematic review with meta-analysis. Sports Med Open, 2023, 9(1): 98-112. [Crossref]
28. Hortobágyi T, Vetrovsky T, Brach J, van Haren M, Volesky K, Radaelli R, et al. Effects of exercise training on muscle quality in older individuals: a systematic scoping review with meta-analyses. Sports Med Open, 2023, 9(1): 41-53. [Crossref]
29. Labata-Lezaun N, Canet-Vintró M, López-de-Celis C, Rodríguez-Sanz J, Aiguadé R, Cuadra-Llopart L, et al. Effectiveness of a multicomponent training program on physical performance and muscle quality in older adults: a quasi-experimental study. Int J Environ Res Public Health, 2022, 20(1): 222-235. [Crossref]
30. Ramirez-Campillo R, Alvarez C, Garcìa-Hermoso A, CelisMorales C, Ramirez-Velez R, Gentil P, et al. High-speed resistance training in elderly women: effects of cluster training sets on functional performance and quality of life. Exp Gerontol, 2018, 110: 216-222. [Crossref]
31. Sales W, Mâcedo S, Gonçalves R, Andrade L, Ramalho C, de Souza G, et al. Use of electrical bioimpedance in the assessment of sarcopenia in the older aldults: a scoping review. J Bodyw Mov Ther, 2024, 39: 373-381. [Crossref]
32. Beaudart C, Bruyère O, Geerinck A, Hajaoui M, Scafoglieri A, Perkisas S, et al. Equation models developed with bioelectric impedance analysis tools to assess muscle mass: a systematic review. Clin Nutr ESPEN, 2020, 35: 47-62. [Crossref]
33. Pus K, Paravlic A, & Šimunič B. The use of tensiomyography in older adults: a systematic review. Front Physiol, 2023, 14: 1213993. [Crossref]
34. Ward L, & Brantlov S. Bioimpedance basics and phase angle fundamentals. Rev Endocr Metab Disord, 2023, 24(3): 381-391. [Crossref]
35. Kumar S, Dutt A, Hemraj S, Bhat S, & Manipadybhima B. Phase angle measurement in healthy human subjects through bio-impedance analysis. Iran J Basic Med Sci, 2012, 15(6): 1180-1184.
36. Pavasini R, Guralnik J, Brown J, di Bari M, Cesari M, Landi F, et al. Short physical performance battery and all-cause mortality: systematic review and meta-analysis. BMC Med, 2016, 14(1): 215-222. [Crossref]
37. Podsiadlo D, & Richardson S. The timed “Up & Go”: a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc, 1991, 39(2): 142-148. [Crossref]
38. Virto N, Río de Frutos X, Muñoz Pérez I, Méndez Zorrilla A, & García-Zapirain B. Gait speed in older adults: exploring the impact of functional, physical and social factors. Retos: nuevas tendencias en educación física, deporte y recreación, 2024, (61): 552-566. [Crossref]
39. Alcazar J, Losa-Reyna J, Rodriguez-Lopez C, AlfaroAcha A, Rodriguez-Mañas L, Ara I, et al. The sit-to-stand muscle power test: an easy, inexpensive and portable procedure to assess muscle power in older people. Exp Gerontol, 2018, 112: 38-43. [Crossref]
40. Bohannon R. Grip strength: an indispensable biomarker for older adults. Clin Interv Aging, 2019, 14: 1681-1691. [Crossref]
41. González-Badillo J, & Sánchez-Medina L. Movement velocity as a measure of loading intensity in resistance training. Int J Sports Med, 2010, 31(5): 347-352. [Crossref]
42. Sánchez-Medina L, & González-Badillo J. Velocity loss as an indicator of neuromuscular fatigue during resistance training. Med Sci Sports Exerc, 2011, 43(9): 1725-1734. [Crossref]
43. Koo B. Assessment of muscle quantity, quality and function. J Obes Metab Syndr, 2022, 31(1): 9-16. [Crossref]
44. Kyle U, Genton L, Hans D, & Pichard C. Validation of a bioelectrical impedance analysis equation to predict appendicular skeletal muscle mass (ASMM). Clin Nutr, 2003, 22(6): 537-543. [Crossref]
45. Cardoso A, Fernandes A, Aguilar-Pimentel J, de Angelis M, Guedes J, Brito M, et al. Towards frailty biomarkers: candidates from genes and pathways regulated in aging and age-related diseases. Ageing Res Rev, 2018, 47: 214-277. [Crossref]
46. Erusalimsky J, Grillari J, Grune T, Jansen-Duerr P, Lippi G, Sinclair A, et al. In search of ‘omics’-based biomarkers to predict risk of frailty and its consequences in older individuals: the FRAILOMIC initiative. Gerontology, 2016, 62(2): 182-190. [Crossref]
47. Cassol I, Ibañez M, & Bustamante J. Key features and guidelines for the application of microbial alpha diversity metrics. Sci Rep, 2025, 15(1): 622-635. [Crossref]
48. Herdman M, Gudex C, Lloyd A, Janssen M, Kind P, Parkin D, et al. Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L). Qual Life Res, 2011, 20(10): 1727-1736. [Crossref]
49. Río X, Guerra-Balic M, González-Pérez A, LarrinagaUndabarrena A, & Coca A. Reference values for SPPB in people over 60 years of age in the Basque Country. Atención Primaria, 2021, 53(8): 102075. [Crossref]
50. de Frutos X, Isla J, Leguina A, Guereño P, Sáez Gomez de Cadiñanos I, & Coca A. Reference values for gait speed in elderly over 65 years of age. Retos, 2022, 45: 936-942. [Crossref]
51. Río X, Larrinaga-Undabarrena A, Coca A, & Guerra-Balic M. Reference values for handgrip strength in the Basque Country elderly population. Biology (Basel), 2020, 9(12): 414-423. [Crossref]
52. Virto N, Río X, Angulo-Garay G, García Molina R, Avendaño Céspedes A, Cortés Zamora E, et al. Development of continuous assessment of muscle quality and frailty in older patients using multiparametric combinations of ultrasound and blood biomarkers: protocol for the ECOFRAIL study. JMIR Res Protoc, 2024, 13: e50325. [Crossref]
53. Ramos C, Gibson G, Walton G, Magistro D, Kinnear W, & Hunter K. Systematic review of the effects of exercise and physical activity on the gut microbiome of older adults. Nutrients, 2022, 14(3): 674-686. [Crossref]
54. Urban S, Chmura O, Wątor J, Panek P, & Zapała B. The intensive physical activity causes changes in the composition of gut and oral microbiota. Sci Rep, 2024, 14(1): 20858. [Crossref]