Open Access | Review
This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.
The rationale for testing drug combinations in aging intervention studies
# Contributed equally to this manuscript.
* Corresponding author: Warren Ladiges
Mailing address: Department of Comparative Medicine, School
of Medicine, University of Washington, Seattle, WA 98195,
USA.
E-mail: wladiges@uw.edu
Received: 07 December 2019 / Accepted: 09 December 2019
DOI: 10.31491/APT.2019.12.001
Abstract
Aging is a complex process driven by seven intertwined pillars that functionally decline with increasing chronologic age. These pillars of aging include stem cell function, mitochondrial function, proteostasis, autophagy, nutrient sensing, metabolism, epigenetic control, and adaptation to stress, macromolecular damage and inflammation. All of the pillars appear to be interconnected such that a change in one, impinges upon others. With so many pillars of aging, it makes drug development to target aging processes equally complex. This leads to the notion that multiple pathways or biological processes need to be targeted to effectively prevent, delay or attenuate aging. The concept of drug combinations as a powerful anti-aging platform is intriguing but has yet to be tested systematically. Insulin function, mTOR (mammalian target of rapamycin) signaling and epigenetic regulation are well-established molecular pathways involved in the pathobiology of aging. Existing drugs that target these pathways include acarbose, rapamycin, and phenylbutyrate, respectively. Acarbose and rapamycin, used as single agents, extend the lifespan of mice. Thus, a cocktail of these drugs with different mechanisms of action would be expected to complement one another and robustly enhance a delay of aging and age-related disease not achievable with mono-therapeutic approaches. Studies to test this concept will be helpful in the development of clinical trials to enhance healthy aging.
Keywords
Aging intervention, drug combinations, glucose regulation, mTOR signaling, epigenetics
Aging processes can be targeted with repurposed drugs
Aging is a complex multifactorial process involving multiple pathways that may need to be targeted to effectively
prevent or slow aging [1]. A number of molecular pathways are well known for regulating lifespan, including insulin signaling, mTOR (mammalian target of rapamycin) signaling, and epigenetic regulation by histone deacetylation (HDAC). Selection of these specifc pathways for a
multiplex approach is based on their well-established roles
in aging and age-related diseases, the fact that they can be
targeted by existing, well-characterized and clinically effcacious drugs, and their distinct roles in aging and agerelated diseases leading to the potential for additive or
synergistic effects when simultaneously therapeutically
targeted ( Figure 1 ).
Insulin signaling is influenced by a number of factors
in an age-related manner. For example, age-related insulin resistance is due in part to increased adiposity and
decreased efficiency of clearing misfolded proteins in
the endoplasmic reticulum vital for proper function of
insulin [2, 3]. The mTOR pathway is sensitive to nutrient intake [4], and in an age-related manner enhances
cell growth and proliferation [5], while suppressing vital
autophagy and proteasome-mediated turn-over activities [6]. Epigenetic dysregulation is implicated in a wide variety of age-related diseases [7]. The two major epigenetic
mechanisms that influence gene expression during aging
are DNA methylation and modification of histones that
package DNA. Among the known histone modifcations,
acetylation has the greatest potential to induce chromatin
unfolding and allow increased transcription. Deacetylases
on the other hand, cause a more condensed, transcriptionally repressed chromatin conformation and become more
active with increasing age [8]. Each of the three systems
has been targeted with individual drugs, but only a few
of these drugs have anti-aging affects, and no single drug
has been shown to target all three pathways. Therefore,
combinations of these drugs and the approach of simultaneously targeting multiple pillars of aging is anticipated
to impact health span and lifespan greater than any single
agent [9].
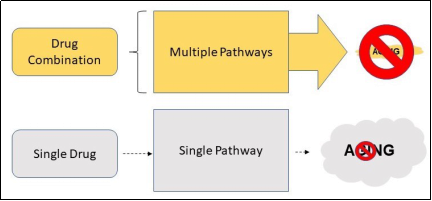
Figure 1. A drug combination that targets multiple pathways of aging has much more potential of suppressing aging than a single drug that only targets a single pathway.
Selection of drug combinations is based on physiological and molecular effects of each individual drug
The criteria for selecting which drugs to target these pathways as a combination approach in mouse studies must be
based on well-validated preclinical evidence of anti-aging
effects of individual drugs either by lifespan extension, for
example by the NIA-supported Intervention Testing Program, or health span enhancement, or both. A relatively
small number of drugs fit these criteria and three drugs
can be identifed to form the basis of a prototype cocktail:
acarbose (Acb) [10], rapamycin (Rap) [11-13] and phenylbutyrate (Pba) [14]. All three are clinically approved
drugs with extensive biological, clinical, and safety data.
Acb has an antihyperglycemic effect resulting from competitive, reversible inhibition of membrane-bound intestinal alpha-glucosidase and pancreatic alpha-amylase, two
enzymes needed to digest complex carbohydrates [15].
In diabetic patients, inhibiting these enzymes results in
delayed glucose absorption and a lowering of postprandial
hyperglycemia. Acb is considered a very safe and effective drug with only a fraction of patients experiencing side
effects consisting of intestinal gas and diarrhea. Acb does
not inhibit lactase and consequently does not induce lactose intolerance. Acb does not affect fasting blood glucose
concentrations significantly or affect insulin secretion,
so it does not cause hypoglycemia when given as monotherapy [16]. However, as a precaution, blood glucose and
glycosylated hemoglobin HbA1c are usually monitored. It
is contraindicated in diabetic ketoacidosis and inflammatory bowel disease. In the NIA Intervention Testing Program, acarbose increased medium and maximal lifespan
in HET3 mice when treatment was started at 4 months
of age [1] and at 16 months of age [17]. Tong et al [18] treated SAMP8 mice with Acb for six months and showed
an alleviation of age-related behavioral and biochemical changes. Acb prevents cardiac ischemia/reperfusion injury
in mice caused by post-prandial hyperglycemia [19].
Rap primarily targets complex 1 of TOR (TORC1), which
is regulated by upstream pathways responsive to nutrient intake such as carbohydrates and amino acids [4].
High nutrients leading to TORC1 activation translates to
rapid cell growth and proliferation, while a rapamycinmediated reduction in TORC1 signaling may phenocopy
dietary restriction, which is known to enhance longevity
in numerous model organisms. Downstream effects of reduced TORC1 signaling include enhanced autophagy and
proteasome-mediated turnover [20, 21]. Rap and derivatives are currently being tested in a wide range of clinical
trials for numerous chronic diseases. Long term rapamycin treatment results in several side effects including hyperglycemia and dyslipidemia, and testicular atrophy [12].
Transient short-term treatment may alleviate these side
effects by delivering less drug for a shorter period of time [22]. A geropathology grading platform [23]was used to
compare lesion scores in C57BL/6 male mice treated with
Rap for two months, starting treatment at 24 months of
age. Micro-encapsulated Rap was delivered in the feed at
42 ppm, and no difference in amount of food ingested in
any of the cohorts was detected over the course of treatment. Mice treated with Rap had signifcantly lower lesion
scores compared to control mice [24,25].
Pba is a broad-spectrum inhibitor of class 1 and class 2
HDACS (both classes have zinc-driven catalytic sites)
and inhibits histone deacetylation such that expression of
genes associated with an overall slowing of cell growth
and proliferation [7]. It is of interest that class 3 HDACS
include the sirtuins, which have an absolute requirement
for NAD+, and are targeted by activators rather than inhibitors such as Pba. Pba is also an active endoplasmic
reticulum (ER) stress chaperone that enhances protein
folding and decreases ER stress [26]. Therefore, there is
rationale to consider Pba as part of a treatment strategy for
heart disease, cancer, neurodegeneration, and inflammation. We have shown that C57BL/6 mice treated with Pba
for 12 months beginning at 4 months of age did not experience cognitive impairment associated with aging [26] and had decreased geropathology lesion scores in multiple
organs (unpublished observations).
A drug cocktail is a promising approach to enhance healthy aging and delay age-related diseases by complementary interacton
The rationale for combining the three drugs as a cocktail
is based on expected mechanisms of molecular and cellular interaction [9]. Oral Acb will block intestinal alphaglucosidase so that carbohydrates are not broken down
and absorbed. This will lower blood glucose levels and
prevent postprandial insulin spikes. The lower blood glucose and decreased need for insulin will activate AMPK,
which blocks mTORC1, the drug target for Rap. The
lowered blood glucose provides less substrate for mitochondrial metabolism thereby stimulating mitochondria
to increase electron transport chain (ETC) efficiency.
The decreased demand for insulin helps alleviate insulin
resistance induced by rapamycin-suppressed mTORC2.
Rap will block mTORC1 signaling resulting in suppression of protein synthesis, suppression of mitochondrial
metabolism, and activation of autophagy. The suppressed
metabolic activity will increase ETC efficiency for ATP
production and decrease the generation of ROS thought to
drive macromolecular damage. The suppression of protein
synthesis will conserve valuable cellular resources. Pba
will activate genes involved in an overall slowing of cell
growth and proliferation and will improve proper insulin
maturation and more effcient insulin signaling. Activation of autophagy will help eliminate damaged macromolecules and organelles. The decreased substrate triggered
by Acb and the downregulation of the mitochondrial
proteasome by Rap will provide a cellular environment
reminiscent of a more youthful phenotype.
In conclusion, drug combinations such as rapamycin, acarbose, and phenylbutyrate are a novel approach designed
to complement mechanisms of action of each individual
molecular target and robustly enhance a delay of aging
and age-related disease not seen with mono-therapeutic
approaches. This type of drug cocktail could serve as a powerful prototype for developing future aging intervention strategies, but rigorous preclinical and clinical studies
are still needed to assure effcacy and safety especially in
older populations.
Declaration
Conflicts of interest
Warren Ladiges is a member of the Editorial Board of Aging Pathobiology and Therapeutics. All authors declare no conflict of interest and were not involved in the journal’s review or desicions related to this manuscript.
References
1. Ladiges W. The quality control theory of aging. 2014.
2. Ladiges W, Morton J, Blakely C, et al. Tissue specific expression of PKR protein kinase in aging B6D2F1 mice. Mechanisms of ageing and development, 2000, 114(2): 123-132.
3. Ladiges W C, Knoblaugh S E, Morton J F, et al. Pancreatic β-cell failure and diabetes in mice with a deletion mutation of the endoplasmic reticulum molecular chaperone gene P58IPK. Diabetes, 2005, 54(4): 1074-1081.
4. Loewith R, Hall M N. Target of rapamycin (TOR) in nutrient signaling and growth control. Genetics, 2011, 189(4): 1177-1201.
5. Vellai T, Takacs-Vellai K, Zhang Y, et al. Genetics: influence of TOR kinase on lifespan in C. elegans. Nature, 2003, 426(6967): 620.
6. Kapahi P, Zid B M, Harper T, et al. Regulation of lifespan in Drosophila by modulation of genes in the TOR signaling pathway. Current Biology, 2004, 14(10): 885-890.
7. Pasyukova E G, Vaiserman A M. HDAC inhibitors: A new promising drug class in anti-aging research. Mechanisms of ageing and development, 2017, 166: 6-15.
8. Walsh M E, Van Remmen H. Emerging roles for histone deacetylases in age-related muscle atrophy. Nutrition and healthy aging, 2016, 4(1): 17-30.
9. Ladiges W, Liggitt D. Testing drug combinations to slow aging. 2018.
10. Harrison D E, Strong R, Sharp Z D, et al. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. nature, 2009, 460(7253): 392.
11. Harrison D E, Strong R, Allison D B, et al. Acarbose, 17-α- estradiol, and nordihydroguaiaretic acid extend mouse lifespan preferentially in males. Aging cell, 2014, 13(2): 273-282.
12. Wilkinson J E, Burmeister L, Brooks S V, et al. Rapamycin slows aging in mice. Aging cell, 2012, 11(4): 675-682.
13. Flynn J M, O'Leary M N, Zambataro C A, et al. Late-life rapamycin treatment reverses age-related heart dysfunction. Aging cell, 2013, 12(5): 851-862.
14. Walsh M E, Bhattacharya A, Sataranatarajan K, et al. The histone deacetylase inhibitor butyrate improves metabolism and reduces muscle atrophy during aging. Aging Cell, 2015, 14(6): 957-970.
15. Balfour J A, McTavish D. Acarbose. An update of its pharmacology and therapeutic use in diabetes mellitus. Drugs, 1993, 46(6): 1025-1054.
16. Yamamoto M, Otsuki M. Effect of inhibition of alphaglucosidase on age-related glucose intolerance and pancreatic atrophy in rats. Metabolism, 2006, 55(4): 533- 540.
17. Strong R, Miller R A, Antebi A, et al. Longer lifespan in male mice treated with a weakly estrogenic agonist, an antioxidant, an α-glucosidase inhibitor or a Nrf2-inducer. Aging cell, 2016, 15(5): 872-884.
18. Tong J J, Chen G H, Wang F, et al. Chronic acarbose treatment alleviates age-related behavioral and biochemical changes in SAMP8 mice. Behavioural brain research, 2015, 284: 138-152.
19. Frantz S, Calvillo L, Tillmanns J, et al. Repetitive postprandial hyperglycemia increases cardiac ischemia/reperfusion injury: prevention by the α-glucosidase inhibitor acarbose. The FASEB journal, 2005, 19(6): 591-593.
20. Powers R W, Kaeberlein M, Caldwell S D, et al. Extension of chronological life span in yeast by decreased TOR pathway signaling. Genes & development, 2006, 20(2): 174-184.
21. Johnson S C, Rabinovitch P S, Kaeberlein M. mTOR is a key modulator of ageing and age-related disease. Nature, 2013, 493(7432): 338-345.
22. Arriola Apelo S I, Neuman J C, Baar E L, et al. Alternative rapamycin treatment regimens mitigate the impact of rapamycin on glucose homeostasis and the immune system. Aging cell, 2016, 15(1): 28-38.
23. Snyder J M, Snider T A, Ciol M A, et al. Validation of a geropathology grading system for aging mouse studies. GeroScience, 2019, 41(4): 455-465.
24. Ladiges W, Snyder J M, Wilkinson E, et al. A new preclinical paradigm for testing anti-aging therapeutics. Journals of Gerontology Series A: Biomedical Sciences and Medical Sciences, 2017, 72(6): 760-762.
25. Bell-Temin H, Yousefzadeh M J, Bondarenko A, et al. Measuring biological age in mice using differential mass spectrometry. Aging (Albany NY), 2019, 11(3): 1045.
26. Wiley J C, Pettan-Brewer C, Ladiges W C. Phenylbutyric acid reduces amyloid plaques and rescues cognitive behavior in AD transgenic mice. Aging Cell, 2011, 10(3): 418-428.