Open Access | Review
This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.
Non invasive techniques for muscle quality functional domain monitoring in older adults: a scoping review
* Corresponding author: Naiara Virto
Mailing address: eVida Research Lab. Faculty of Engineering. University of Deusto, Physical Activity and Sport Science Department, Faculty of Health Sciences, University of Gasteiz - EUNEIZ, Vitoria-Gasteiz, Spain.
Email: naiara.virto@euneiz.com
This article belongs to the Special Issue: Healthy Aging: Physical activity intervention
Received: 11 February 2026 / Revised: 24 February 2026 / Accepted: 20 March 2026 / Published: 31 March 2026
DOI: 10.31491/APT.2026.03.204
Abstract
Background:The EWGSOP update emphasizes the importance of assessing muscle quality (MQ) as a key indicator of skeletal muscle function. MQ defined by functional domains, reflects how effectively a muscle generates force relative to its size, often quantified through muscle quality index. Age-related declines in MQ typically precede loss of muscle mass, highlighting its potential as a more sensitive functional marker. This review focuses on indirect non-invasive techniques for assessing the functional domain of MQ, which are practical, accessible, and widely applicable in older populations. Therefore, the aim is to map and synthesise current evidence on non-invasive tools to assess functional MQ in older adults as well as to promote the adoption of accessible and simple methods in community settings.
Results:A total of 79 studies were included. The definitions align with indirect measures of muscle function relative to muscle mass (strength per unit of muscle mass). For strength assessment, key tools included handheld dynamometers, isokinetic dynamometers, leg power tests, RM testing, and combined strength measures. For muscle mass, methods included dual energy Xray absorptiometry (DXA), ultrasound, bioelectrical impedance analysis (BIA), magnetic resonance imaging (MRI), and computed tomography (CT).
Conclusions:This review highlights the heterogeneity in MQ, particularly in the methods used to assess muscle strength and mass. A variety of non-invasive tools are available for assessing MQ, with handheld dynamometers and 1RM tests being practical for community use, while isokinetic dynamometers and power assessments are more specialized. For muscle mass, MRI, DXA, and CT are widely used to assess structural and compositional aspects of muscle but costly, whereas BIA and ultrasound offer more accessible and cost-effective alternatives for community settings.
Keywords
Muscle quality, functional domain, non invasive techniques, older adults, assessment methods
Introduction
The aging process induces neural and morphological changes in the human musculoskeletal system, leading to a decline in muscle function and structure. These changes result in a progressive reduction in muscular strength, mass, and quality, primarily due to the natural degeneration of muscle, bone, and joint tissues that accompanies aging [1]. These decreases in muscular parameters reflect both qualitative and quantitative alterations in skeletal muscle structure and function, which can lead to sarcopenia [2]. According to European Working Group on Sarcopenia in Older People (EWGSOP), sarcopenia is a gradual, widespread skeletal muscle condition that is linked to a higher risk of experiencing negative health consequences such as falls, fractures, physical impairment, frailty, and death [2].
The latest EWGSOP update, as well as other recognised working groups, take to the forefront the importance of assessing muscle quality (MQ) as a crucial indicator of the physiological properties of skeletal muscle tissue, which influence both muscle strength and functional capacity [2]. The term “muscle quality” describes the physiological potential and functional performance of muscular tissue. MQ involves various aspects of muscle composition, including both microscopic and macroscopic features of muscle structure, along with functional performance relative to muscle mass [2, 3]. For these reasons, it is typically defined by two primary domains: morphological and functional. The morphological domain involves direct evaluations of muscle architecture, considering both microscopic and macroscopic characteristics of muscle structure, as well as its composition [2-4]. In contrast, the functional domain involves indirect evaluations of muscle performance relative to its mass. This domain aims to measure how effectively a muscle generates force in relation to its size, often represented by a MQ index [3, 5, 6]. Assessing MQ from this functional standpoint involves examining key performance metrics, such as muscle performance, taking into account the type and direction of the movement, such as strength and power (including peak torque (PT), 1 repetition maximum (RM), 5RM, isometric strength, and others) alongside quantitative parameters of muscle size, including muscle mass, lean mass, muscle thickness, muscle volume and/or cross-sectional area [3, 5, 6].
Age-related alterations in MQ tend to occur before any noticeable decline in muscle mass, indicating that MQ might serve as a more sensitive indicator of muscle function than muscle mass alone [3]. This finding is significant, as shifts in MQ could offer a clearer explanation for the declines in muscle strength and function commonly seen with aging [7]. In this regard, maintaining and assessing MQ becomes essential for older adults in order to slow down changes in muscle metabolism and assist avoid the loss of muscle mass and strength [8]. Additionally, it is important to take into account that MQ is a relatively recent term, and the diagnostic methods for evaluating it are not yet fully standardized [2], underscoring the need for further research in this field.
MQ assessment methods can be divided into three primary categories: first, invasive techniques that involve direct intervention; second, direct non-invasive techniques; and third, indirect non-invasive methods, which evaluate MQ through external measures or estimates [2-4]. Specifically, this scoping review will focus on indirect techniques that measure the functional domain of MQ. These techniques are relatively easy to perform, do not require the insertion of invasive devices and are an important tool for assessing functional MQ in older adults [5, 6, 9].
To assess functional MQ, commonly used tools are designed to measure both strength or power and muscle mass. These measurements are crucial as they form the basis for calculating the MQ index, estimated by indirect assessments of muscle performance in relation to muscle mass [3, 5, 6]. In terms of measurement tools for assessing MQ, each has specific applications in clinical practice. For measuring muscle strength, simpler tools such as isometric dynamometers and RM strength tests can be used, while more advanced devices such as power gauges and isokinetic strength testers offer more detailed assessments [2, 10, 11]. For muscle mass, there are several techniques: Bioelectrical Impedance Analysis (BIA), Dual energy Xray absorptiometry (DXA), B-Mode Ultrasound, Computed Tomography (CT) and Magnetic Resonance Imaging (MRI). Each tool provides different information on muscle mass, capturing measures such as muscle mass, lean mass, muscle thickness, muscle volume or cross-sectional area [9, 12, 13].
Institutions such as ICFSR and EWGSOP highlight the need for standardized definitions and reliable methods to assess MQ. However, effective evaluation techniques remain unclear, emphasizing the importance of reviews that compile and analyze current evidence on functional MQ assessment methods [2, 9]. This scoping review aims to map and synthesize current evidence on non-invasive tools for assessing functional MQ in older adults, identifying methods currently used in the literature, trends, and advances. It also examines tool limitations and factors influencing their selection, including accessibility, cost, and training, to promote simple, community-based applications.
Methods
Registration
The scoping review was registered on the Open Science Framework (OSF) platform in which the search strategy and supplementary materials are available. ( https://osf.io/ anjr4/?view_only=05969c336a0847028766e96f574eb6 3e), on November 4, 2024 (registration DOI:https://doi. org/10.17605/OSF.IO/QV28T).
Procedures
The review was carried out in accordance with the 2020 preferred reporting items for systematic reviews and metaanalyses (PRISMA) standards.
Research questions
This scoping review aims to address the following research questions: 1. Which are the non-invasive tools and their principles used to measure functional domain of MQ in older adults? 2. Which non-invasive tools are commonly used and reported in the literaturefor assessing the functional domain of MQ in geriatric populations? 3. What are the main limitations of current non-invasive tools for measuring functional domain MQ in older adults? 4. What factors, such as cost, accessibility, and required training, influence the choice of non-invasive tools for assessing functional MQ, particularly in community settings, and how can accessible and simple methods be implemented to facilitate their use in the community? What recent trends or advances have been observed?.
Eligibility criteria
PICOS (participants, interventions, comparators, outcomes, and study design) was used to include and exclude original, peer-reviewed, full-text studies. Table 1 provides a summary of the selection criteria.
Table 1.
Eligibility criteria.
| Category | Inclusion criteria | Exclusion criteria |
|---|---|---|
| Participants | Healthy older adults (+ 60 years) | Studies that included participants under 60 years of age and problems of muscle diseases that may influence the measurement of muscle quality |
| Interventions | Any kind of intervention or lack of intervention | Not applicable |
| Comparators | Testing procedures used for muscle quality functional domain assessment | Studies without assessments of muscle quality in the functional domain |
| Outcomes | Metrics indicating functional domain muscle quality status. Also, psychometric properties were extracted when reported in the included studies | Muscle quality asses directly (morphological domain) or invasively |
| Study design | Randomized controlled and non-randomized controlled trials, observational studies and one group studies | Systematic reviews and meta-analysis |
Literature search and screening process
The initial search strategy was developed by one reviewer (NV) specifically for PubMed (outlined below), targeting title, abstract, and keyword fields. This strategy was subsequently adapted to conform with the syntax and subjectspecific headings of additional databases. A comprehensive literature search was then executed in PubMed, Web of Science, and Scopus databases, employing Boolean operators to combine relevant keywords systematically (aged OR old OR elder* OR aging OR frail* OR older OR senior OR geriatric) AND (measure* OR determine OR analyze OR evaluate OR “sensitivity and specificity” OR screening OR tool* OR assess* OR “diagnostic accuracy”) AND (“muscle function” OR “muscle performance” OR “muscle functional performance” OR “muscle functional capacity” OR “muscle quality” OR “functional domain” OR “muscle quality index”). The search was performed without date restriction and was updated until October 2024.
An initial search was conducted by one author (NV), and records were uploaded to Rayyan QCRI to remove duplicates. Two reviewers (NV and XR) independently screened titles and abstracts, resolving discrepancies collaboratively. Full texts were assessed using predefined criteria, with exclusions recorded. Non-invasive imaging modalities were not used as search terms in the search strategy to avoid biasing the retrieval of studies toward specific measurement techniques. The search instead focused on the concept of MQ and its functional assessment.
Nevertheless, studies using these methods were included during the screening process when they reported functional MQ indices.
Data collection
Data from the included studies were collected and coded in Microsoft Excel (Microsoft Corp). The following information was extracted from each included study: (1) reference, author, year of publication and country, (2) participants characteristics (sample size; sex; age (mean and standard deviation) and health status), (3) muscle quality definition, (4) muscle quality assessment methods (test and instruments; principles of measurement), (5) group of muscles on which the measurement has been performed, and (6) reported advantages and limitations of the measurements tools. In addition to extracting the operational definition of MQ reported in each study, all metrics were categorised according to the physiological construct they primarily represented. Based on previous conceptual descriptions of MQ, the identified indicators were grouped into three categories: (1) strength-to-mass ratios, defined as muscle strength normalised to a measure of muscle or body mass; (2) strength-to-morphology ratios, in which muscle strength was normalised to a muscle-specific morphological parameter obtained through imaging techniques such as cross-sectional area, muscle thickness or echo intensity; and (3) field-test surrogate measures, which estimate MQ indirectly through functional performance tests.
Results
Study selection
Potential studies were directly exported from scientific databases into Rayyan (https://www.rayyan.ai/) for the removal of duplicates and subsequent screening based on the predefined inclusion and exclusion criteria. After completing these steps, a total of 10,559 records were identified. The study selection process is outlined in Figure 1. Duplicates were removed (n = 6,591), and following the screening of titles and abstracts, 6,382 records were excluded. This left 209 full-text articles for further evaluation. Of these, an additional 130 studies were excluded after full-text review for eligibility. Ultimately, 79 studies were deemed eligible for inclusion in the scoping review.
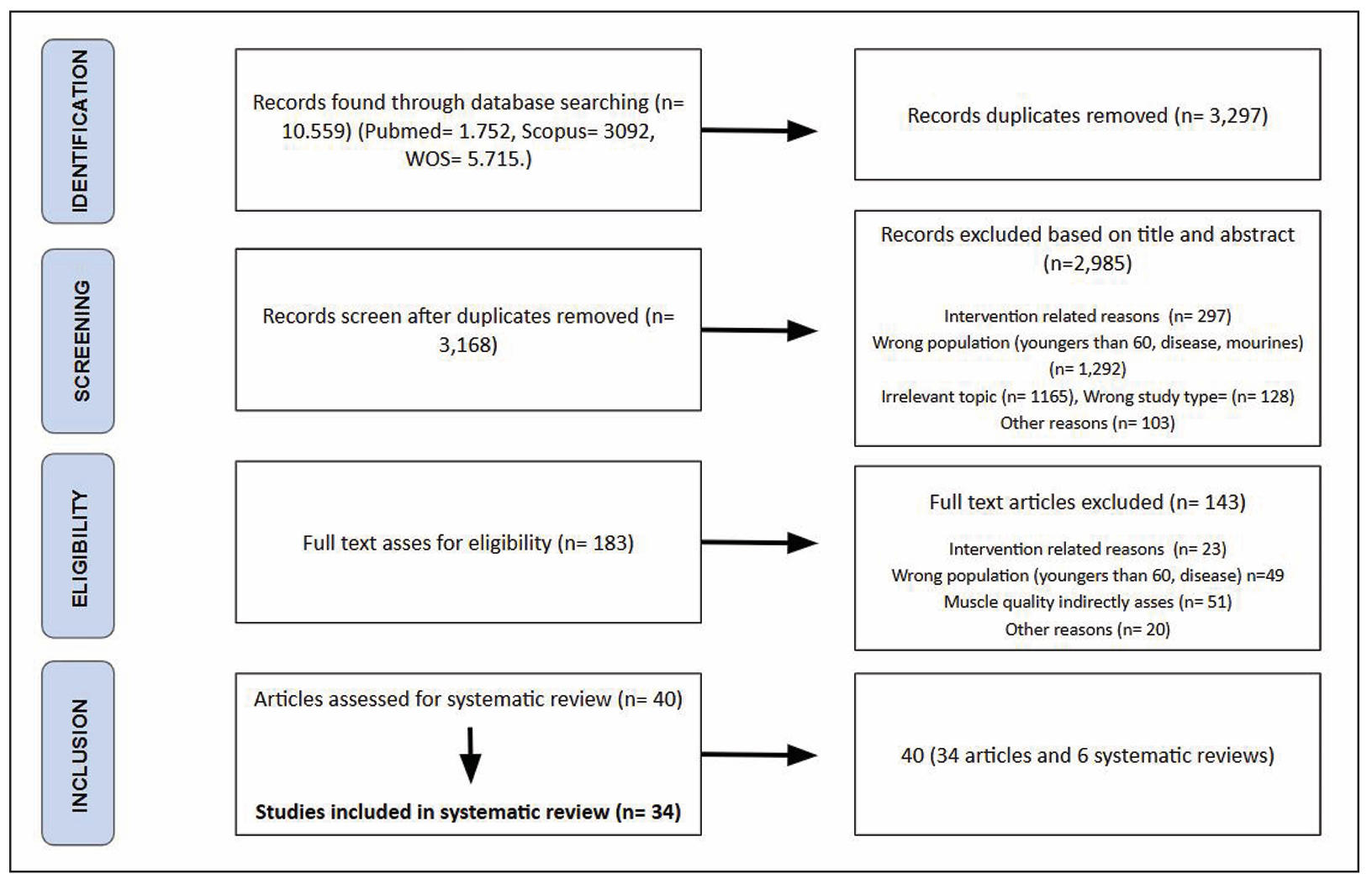
Figure 1. Flow diagram of the systematic search process
Study characteristics
The characteristics of the included studies are detailed in Table 2 and a subset of representative studies illustrating the different operational definitions of MQ characteristics is detailed in Table S1. A total of 32,892 participants, with an age older than 60 years, were analyzed in this review. Regarding participants sex, 42 studies reported a sample consisting of both males and females (n = 26,572, 80.8% of total participants). 30 studies were composed of only females (n = 3,719, 11.3% of total participants) and 7 groups involved only males(n = 2,601, 7.9% of total participants). The selected studies were published between 1999 and 2024, with approximately a third being published since 2021 (n = 24, 30%).
Table 2.
Descriptive characteristics of participants, definition used for muscle quality, testing procedures, muscle group and reported advantages and limitations of the measurement tools.
| Country | Reference | Participants | Muscle quality definition and category | Assessment methods (test and principles of measurement) | Assessment anatomical site | Reported advantages and limitations |
|---|---|---|---|---|---|---|
| Brazil | Abdalla et al. [14] | 94 community-dwelling older adults (69.1% women, 69 ± 6.1 yrs) | Strength-to-mass ratio. Muscle quality ratio standard (muscle strength normalized by body size). |
Test: HGS, knee extensors (1RM), isokinetic knee extension peak torque. Instrument: for muscle strength: isometric dynamometer (HGS), isokinetic dynamometer (lower limbs muscle), for muscle mass: DXA and BIA. |
Lower and upper limbs (leg and arm). | HGS and BIA are accessible and inexpensive, ideal for community and clinical use, but there are more specific imaging techniques. Isokinetic dynamometer and DXA are commonly used for assessing leg strength and mass, but are costly and require specialized equipment and training, limiting use in non-clinical settings. |
| Japan | Abe et al. [15] | 135 active elderly males (77 ± 4 yrs) | Strength-to-morphology ratio. Forearm muscle quality, ratio of HGS to forearm ulna muscle thickness (HGS/MT). Relative HGS ratio of HGS to forearm girth (HGS/forearm girth) and a ratio of HGS to body mass (HGS/body mass). |
Test: HGS. Instrument: for muscle strength: isometric dynamometer (HGS), for muscle thickness: B-mode US. |
Upper limbs (forearm). | These types of dynamometers are used to assess functional muscle quality and feasible for community programme and ultrasound allows detailed assessment of muscle quality, though high cost and operator expertise limits their community accessibility. |
| South Korea | Baek et al. [16] | 143 older adults (69% women, mean age 74 ± 6 yrs) | Strength-to-mass ratio. Handgrip strength per unit of body weight. |
Test: HGS. Instrument: for muscle strength: isometric dynamometer (HGS), for muscle mass: BIA (body composition). |
Upper limbs (arm). | HGS and BIA are accessible and inexpensive, ideal for community and clinical use, but there are more specific imaging techniques. |
| Iran | Banitalebi et al. [17] | 63 women, osteosarcopenic obesity (65-80 years) | Field-test surrogate measure MQ formula: (Leg Length × 0.4 body mass × gravity × 10/time sit-stand). |
Test: HGS and 30 second chair stand test. Instrument: for muscle strength: isometric dynamometer (HGS), for body composition: DXA. |
lower limbs (leg). | Handgrip and DXA assessments are reliable for community use, though DXA's expense and equipment needs can limit broader accessibility. |
| Canada | Barbat et al. [18] | 1219 women (80 ± 4 yrs) | Strength-to-mass ratio. Muscle quality index (MQI): muscle strength per unit of muscle mass. Upper limb: handgrip strength by upper limbs muscle mass (kPa/kg). Lower limb: knee extension strength (KES) by lower limbs muscle mass (kPa/kg). |
Test: HGS and knee extension strength (KES) test. Instrument: for muscle strength: isometric dynamometer (HGS) and maximum voluntary contraction for (KES), for muscle mass and body composition: DXA, |
Lower and upper limbs (leg and arm). | MQI is more accessible, but may not capture the full spectrum of muscle quality captured by tools such as MRI or CT. DXA assessments are reliable for community use, though DXA's expense and equipment needs can limit broader accessibility. |
| USA | Brady et al. [19] | 94 community-dwelling older women (73.6 ± 5.4) | Strength-to-mass ratio. MQ, leg power normalized for lower-body mineral-free lean mass (watts/kg). |
Test: leg extension power. Instrument: for leg extension power: nottingham power rig, for body composition: DXA. |
Lower limb (quadriceps muscle). | DXA is widely used and often considered a reference method for muscle mass assessment, but high-tech equipment of both limits accessibility. |
| USA | Briggs et al. [20] | 17 older adults post-hip fracture (mean age 77 ± 12 yrs) | Strength-to-mass ratio. MQ, isometric peak knee extension force by the quadriceps muscle lean muscle mass (N/cm2). |
Test: knee extension isometric force. Instrument: for muscle strength: isokinetic dynamometer, for muscle mass: MRI. |
Lower limb (quadriceps muscle). | MRI effective but costly and complex for community implementation. |
| USA | Brightwell et al. [21] | 23 healthy sedentary to low-active older adults (73 ± 4 yrs) | Strength-to-mass ratio. MQ, strength per unit muscle mass. Isokinetic peak torque of right leg divided by right leg lean mass (torque/kg). |
Test: isokinetic (120°/s) peak, torque strength. Instrument: for muscle strength: isokinetic dynamometer, for body composition: DXA. |
Lower limb (quadriceps muscle). | Isokinetic dynamometer and DXA are commonly used for assessing leg strength and mass, but are costly and require specialized equipment and training, limiting use in non-clinical settings. |
| USA | Brown et al. [22] | 4510 older adults in a longitudinal cohort study (≥ 60 yrs) | Field-test surrogate measure. MQI: based on sit-to-stand time, body mass, and leg length. [(L - 0.5) × body mass × g × 5=]/Tsit to stand, where 0.5 (m), Leg (m), g (m/s2). |
Test: 5-repetition sit-to-stand. Instrument: for functional strength: anthropometry for leg length and digital scale for body mass. |
Lower limbs. | An accessible index predicting mortality, though less precise for comprehensive MQ. May quantify age-related decline in locomotion, essential for physical function and longevity, highlighting the need for further research to complement other measures. |
| USA | Canon et al. [23] | 867 (445 females) older adults in a longitudinal cohort study (≥ 60 yrs) | Strength-to-mass ratio. MQ, isokinetic strength per unit muscle mass. Knee extensor strength/ leg skeletal muscle mass. |
Test: knee extensor isokinetic strength. Instrument: for muscle strength: Kin-Com dynamometer (isokinetic strength), for muscle mass: DXA. |
Lower limbs. | Isokinetic dynamometer and DXA are commonly used for assessing leg strength and mass, but are costly and require specialized equipment and training, limiting use in non-clinical settings. |
| Taiwan | Chang et al. [24] | 180 older adults with T2DM (72.5 ± 5.3 yrs, 53.3% males) | Strength-to-mass ratio. MQ, handgrip strength divided by relative appendicular muscle mass. |
Test: HGS. Instrument: for muscle strength: isometric dynamometer (HGS), for muscle mass and body composition: DXA. |
Upper limbs. | Handgrip and DXA assessments are reliable for community use, though DXA's expense and equipment needs can limit broader accessibility. |
| Brazil | Correa et al. [25] | 10 sedentary elderly women (67 ± 5 years) | Strength-to-morphology ratio. MQ, force per unit of muscle mass. The 1RM value of the dominant leg muscle volume of the dominant leg (kg/cm3). |
Test: 1RM test for dominant leg. Instrument: 1RM knee extension dynamometers for strength, for muscle volume B-mode US. |
Lower limb (quadriceps muscle). | 1RM test and ultrasound are frequently reported for precise assessments of muscle quality, though US high cost and operator dependence limits their community accessibility. |
| Brazil | Costa Pereira et al. [26] | 176 hospitalized older adults (≥ 60 yrs, 43.8% women) | Strength-to-mass ratio. MQI: ratio of muscle strength to appendicular skeletal muscle mass (ASMM). |
Test: HGS. Instrument: for muscle strength: isometric dynamometer (HGS), for muscle mass assessment BIA. |
Upper limbs. | HGS and BIA are accessible and inexpensive, ideal for community and clinical use, but there are more specific imaging techniques. |
| Multiple | Cramer et al. [27] | 330 malnourished older adults with sarcopenia (≥ 65 yrs) | Strength-to-mass ratio. Leg strength (PT) relative to leg muscle mass (Nm/kg). |
Test: knee extension. Instrument: for muscle strength: maximal voluntary isokinetic peak torque leg extension, for muscle mass: DXA. |
Lower limbs. | Isokinetic dynamometer and DXA are commonly used for assessing leg strength and mass, but are costly and require specialized equipment and training, limiting use in non-clinical settings. |
| Brazil | Cunha et al. [28] | 62 women (68.6 ± 5.0 yrs) | Strength-to-mass ratio. MQ, specific force per unit muscle mass. Sum of the best 1RM values for the 3 exercises by the skeletal muscle mass. |
Test: knee extension, chest press and preacher curl. Instrument: for muscle strength: sum of 1RM of three exercises, for total skeletal muscle mass: DXA. |
Lower and upper limbs (leg and arm). | These types of dynamometers are used to assess functional muscle quality and feasible for community programmes, while DXA provides a detailed assessment of mass, but is limited to clinical settings due to its cost. |
| USA | Delmonico et al. [29] | 1367 well-suited | Strength-to-morphology ratio. MQ, knee extensor torque per unit of thigh cross sectional area. |
Test: knee extensor torque. Instrument: for muscle strength, Kin-Com dynamometer for isokinetic strength, CT for CSA measurement. |
Lower limbs. | Isokinetic dynamometry and CT offer detailed assessments but are expensive and less feasible for broad application. |
| Brazil | Dos Santos et al. [30] | Functioning older adults (726 men, 70–79 yrs) | Strength-to-mass ratio. Total strength (sum of the exercises tested in the 1RM tests) relative to skeletal muscle mass. |
Test: knee extension, chest press and preacher curl. Instrument: for muscle strength: sum of 1RM of three exercises, for total skeletal muscle mass: DXA. |
Lower and upper limbs (leg and arm). | These types of dynamometers are used to assess functional muscle quality and feasible for community programmes, while DXA provides a detailed assessment of mass, but is limited to clinical settings due to its cost. |
| USA | Emerson et al. [31] | 34 sarcopenic older women (> 60 yrs) | Strength-to-mass ratio. Grip strength relative to appendicular lean mass. |
Test: HGS. Instrument: for muscle strength: isometric dynamometer (HGS), for muscle mass and body composition: DXA. |
Upper limb. | Handgrip and DXA assessments are reliable for community use, though DXA's expense and equipment needs can limit broader accessibility. |
| USA | Dalle et al. [32] | 58 healthy older adults (71 ± 6 yrs, 33 women) | Strength-to-mass ratio. Isometric strength per unit of muscle mass. |
Test: Isometric knee extension. Instrument: for muscle strength: isometric dynamometer, for muscle mass and body composition: CT. |
Lower limb. | CT provides accurate assessment, but requires specialized equipment, limiting its accessibility in community settings. These types of dynamometers are used to assess functional muscle quality and feasible for community programmes. |
| USA | Fragala et al. [33] | 23 community-dwelling, non sarcopenic older adults (65-83 yrs) | Strength-to-mass ratio. MQ, ratio of strength to body mass. Leg extension strength/ lean quadriceps muscle muscle mass (kg/kg). |
Test: knee extension. Instrument: for muscle strength: PLLE power lift knee extension, machine for muscle mass and body composition: DXA. |
Lower limbs. | DXA is widely used and often considered a reference method for muscle mass assessment, but high-tech equipment of both limits accessibility. |
| USA | Fragala et al. [34] | 23 older adults (61-85 yrs) | Field-test surrogate measure. Muscle quality index (MQI) based on sit-to-stand power, body mass, and leg length. MQI = ((leg length × 0.4) × body mass × gravity × 10)/time sit to stand. |
Test: 5 Sit-to-stand (STS), leg length. Instrument: STS performance, for body mass and leg length: DXA. |
Lower limbs. | MQI is practical for community settings, is sensitive and reliable to detect improvements, reflects functional muscle quality and is a potential use for the clinic and community care. However, DXA's expense and equipment needs can limit broader accessibility. |
| Brazil | Gadelha et al. [35] | 25 older adults (70.6 ± 6.1 yrs) | Strength-to-morphology ratio. PT per unit of thigh muscle thickness. |
Test: knee extension. Instrument: isokinetic dynamometer for dominant leg strength, B-mode ultrasound for muscle thickness. |
Lower limb (dominant leg thigh). | Isokinetic dynamometer and ultrasound allow assessment of muscle quality, though high cost and operator expertise limits their community accessibility. |
| Brazil | Gadelha et al. [36] | 246 community-dwelling older women (68.1 ± 6.2 yrs) | Strength-to-morphology ratio. Ratio of PT to thigh muscle thickness. |
Test: knee extension. Instrument: isokinetic dynamometer for dominant leg strength, B-mode ultrasound for muscle thickness. |
Lower limb (dominant leg thigh). | Isokinetic dynamometer and ultrasound allow assessment of muscle quality, though high cost and operator expertise limits their community accessibility. |
| Germany | Ghasemikaram et al. [37] | 167 older women (68.1 ± 6.2 yrs) | Strength-to-mass ratio. 1. Maximum isokinetic hip/leg extensor strength (MILES) per unit of mid-thigh intra-fascia volume. 2. MILES/ thigh muscle mass. |
Test: knee extension. Instrument: for muscle strength: isokinetic leg press, for muscle volume: MRI, for muscle mass: DXA. |
Lower limb. | MRI provides precise volume measures but is expensive and less accessible; isokinetic dynamometer and DXA are providing information for assessing leg strength and mass, but are costly and require specialized equipment and training, limiting use in non-clinical settings. |
| USA | Goodpaster et al. [38] | 43 older men with osteosarcopenia (78 ± 4 yrs) | Strength-to-mass ratio. Isokinetic torque per unit of leg lean mass (Nm/kg). |
Test: knee extensor isokinetic strength. Instrument: for muscle strength: Kin-Com dynamometer (isokinetic strength), for muscle mass: DXA. |
Lower limbs. | Isokinetic dynamometer and DXA are commonly used for assessing leg strength and mass, but are costly and require specialized equipment and training, limiting use in non-clinical settings. |
| Australia | Hairi et al. [39] | 1880 older adults (73.5 ± 2.8 yrs, 51.6% women) | Strength-to-mass ratio. MQ, ratio of strength per unit of mass in lower and upper extremities. |
Test: HGS and quadriceps muscle strength. Instrument: for muscle strength: Jamar isometric dynamometer, spring gauge on both legs separately (lower limbs muscle), for muscle mass: DXA. |
Upper and lower limbs (quadriceps muscle). | Handgrip and DXA assessments are reliable for community use, though DXA's expense and equipment needs can limit broader accessibility. |
| USA | Herda et al. [40] | 1705 older men (≥ 70 yrs) | Strength-to-mass ratio. Upper and lower body muscle strength (1RM) relative to respective lean mass. |
Test: 5RM bench press and 5RM leg press. Instrument: for strength: bench press, standard free-weight bench and half-rack, leg press plate-loaded hip sled with a 45° incline, for lean mass: DXA. |
Upper and lower limbs. | These types of dynamometers are used to assess functional muscle quality and feasible for community programmes, while DXA provides a detailed assessment of mass, but is limited to clinical settings due to its cost. |
| Chile | Jerez-Mayorga et al. [41] | 65 healthy older adults (66.5 ± 7.09 yrs) | Field-test surrogate measure. Muscle Quality Index (MQI) based on sit-to-stand power, body mass, and leg length. MQI = ((leg length × 0.4) × body mass × gravity × 10)/time sit to stand. |
Test: 5 Sit-to-stand (STS), leg length. Instrument: STS performance, for body mass: BIA, leg length was measured manually, applying the anthropometric measurement protocol. |
Lower limbs. | MQI is practical for community settings, is sensitive and reliable to detect improvements, reflects functional muscle quality and is a potential use for the clinic and community care. However, there are more specific imaging techniques. |
| USA | Katsiaras et al. [42] | 28 older women (66.2 ± 5.6 yrs) | Strength-to-morphology ratio. Muscle-specific torque, that is, peak torque per cross-sectional area, was calculated as an index of muscle quality. |
Test: knee extensors and flexors. Instrument: for muscle strength: isokinetic dynamometer in knee extensors and flexors, for CSA: CT. |
Lower limbs. | Isokinetic dynamometry and CT offer detailed assessments but are expensive and less feasible for broad application. |
| Belgium | Kennis et al. [43] | 1512 older adults (70–79 yrs) | Strength-to-morphology ratio. MQ, the ratio of muscle strength to muscle volume (Nm/cm³). 1. MQ isometric strength/vol 2. MQ concentric strength/vol |
Test: knee extensors. Instrument: for muscle strength, isokinetic and concentric strength of the knee extensors was unilaterally measured on a Biodex Medical System 3 dynamometer, for muscle volume: CT. |
Upper and lower limbs (separately). | Isokinetic dynamometry and CT offer detailed and validated assessments but are expensive and less feasible for broad application. |
| Japan | Koji et al. [44] | 72 older men (60-80 yrs) | Strength-to-mass ratio. Leg muscle ratio of strength relative to mass (kg/kg). |
Test: knee extension. Instrument: for muscle strength, hand-held dynamometer was placed proximal to the ankle to measure maximal isometric muscle contraction, for muscle mass: BIA. |
Lower limbs. | HGS and BIA are accessible and inexpensive, ideal for community and clinical use, but there are more specific imaging techniques. |
| USA | Koster et al. [45] | 170 older women (aged 65-79 yrs) | Strength-to-mass ratio. MQ, muscle strength divided by leg lean mass (Nm/kg). |
Test: knee extensor isokinetic strength. Instrument: for muscle strength: Kin-Com dynamometer (isokinetic strength), for muscle mass: DXA. |
Lower limbs. | Isokinetic dynamometer and DXA are commonly used for assessing leg strength and mass, but are costly and require specialized equipment and training, limiting use in non-clinical settings. |
| Taiwan | Liao et al. [46] | 2307 well functioning | Strength-to-mass ratio. 1. MQ upper extremity, handgrip strength by arm lean mass (kg/kg). 2. MQ lower extremity, strength of isometric quadriceps muscle by leg lean mass (N/kg). |
Test: HGS and knee extension. Instrument: for muscle strength: isometric dynamometer (HGS), handheld dynamometer for leg strength, for muscle mass assessment: DXA. |
Upper and lower limbs (separately). | Handgrip and DXA assessments are reliable for community use, though DXA's expense and equipment needs can limit broader accessibility. |
| Brazil | Lopez et al. [47] | older adults | Strength-to-morphology ratio. 1. MQ isometric: ratio between maximum voluntary isometric contraction (MVIC) and plantar flexor muscle thickness. 2. MQ dynamic: ratio between plantar flexor muscle strength at 30°/sec and plantar flexor muscle thickness. |
Test: plantar flexor isometric and dynamic strength. Instrument: for muscle strength, isokinetic dynamometer (MVIC) and dynamic assessment of plantar flexor muscle strength performed at 30°/sec, B-mode ultrasound for muscle thickness. |
Lower limbs. | Isokinetic dynamometer and ultrasound allow assessment of muscle quality, though high cost and operator expertise limits their community accessibility. |
| France | Maltais et al. [48] | 70–79 yrs | Strength-to-mass ratio. 1. MQ, handgrip strength per unit of body weight (kg/kg). 2. MQ, handgrip strength divided by arm fat free mass (kg/kg). |
Test: HGS. Instrument: for muscle strength: isometric dynamometer (HGS), for muscle mass: DXA (body composition). |
Upper limbs (arm). | Handgrip and DXA assessments are reliable for community use, though DXA's expense and equipment needs can limit broader accessibility. |
| USA | McCormack et al. [49] | 56 older women (67.3 ± 5.1 yrs) | Strength-to-mass ratio. MQ, relative strength with grip strength normalized to arm lean mass (kg/kg). |
Test: HGS. Instrument: for muscle strength: isometric dynamometer (HGS), for arm muscle lean mass: DXA (body composition). |
Upper limbs (arm). | Handgrip and DXA assessments are reliable for community use, though DXA's expense and equipment needs can limit broader accessibility. |
| France | Michel et al. [50] | 24 older women (66.3 ± 5.8 yrs) | Strength-to-mass ratio. MQ, ratio of maximum strength normalized to fat-free mass (Nm/kg). |
Test: knee extension. Instrument: for muscle strength: Isokinetic dynamometer in knee extensor, for fat free mass: Sergi equation. |
Lower limbs. | Isokinetic dynamometers are commonly used for assessing leg strength and mass, but are costly and require specialized equipment and training, limiting use in non-clinical settings. |
| USA | Misic et al. [51] | 1679 older adults | Strength-to-mass ratio. MQ, leg strength (summation of peak extension and flexion torque measured by isokinetic dynamometer) normalized for leg mineral free lean mass. |
Test: isokinetic leg extension and flexion. Instrument: for muscle strength: isokinetic dynamometer, for body composition: DXA. |
Lower limbs. | Isokinetic dynamometer and DXA are commonly used for assessing leg strength and mass, but are costly and require specialized equipment and training, limiting use in non-clinical settings. |
| USA | Moore et al. [52] | (75.3 ± 4.4 yrs) | Strength-to-morphology ratio. MQ, knee-extension strength per muscle cross-sectional area. |
Test: knee extensors. Instrument: for muscle strength, isokinetic dynamometer in knee extensors and flexors, for CSA: CT. |
Lower limbs. | Isokinetic dynamometry and CT offer detailed assessments but are expensive and less feasible for broad application. |
| Germany | Müller et al. [53] | 60 apparently healthy older adults (70.7 ± 6.2 yrs) | Strength-to-morphology ratio. MQ, specific tension formula, ratio between the knee extension 1RM/2 and the squared quadriceps muscle femoris muscles (N/cm²). |
Test: knee extension. Instrument: for muscle strength: 1RM test in knee extension, for quadriceps muscle femoris volume B-mode ultrasound derived muscle thickness. |
Lower limbs (quadriceps muscle). | 1RM test and ultrasound allows assessment of muscle quality, though US high cost and operator dependence limits their community accessibility. |
| Brazil | Neri et al. [54] | 184 older adults (73.6 ± 6.83 yrs, 81% women) | Strength-to-mass ratio. MQ, muscle strength for unit of muscle mass. Specific torque (ratio of knee extension peak torque to leg lean mass of the same limb (Nm/kg). |
Test: knee extensors. Instrument: for muscle strength: isokinetic dynamometer, for lean mass: DXA. |
Lower limbs. | Isokinetic dynamometer and DXA are commonly used for assessing leg strength and mass, but are costly and require specialized equipment and training, limiting use in non-clinical settings. |
| Brazil | Neri et al. [55] | 55 healthy older adults (69.3 ± 5.5 yrs) | Strength-to-mass ratio. MQ, muscle strength for unit of muscle mass. Specific torque (ratio of knee extension peak torque to leg lean mass of the same limb (Nm/kg). |
Test: knee extensors. Instrument: for muscle strength: isokinetic dynamometer, for lean mass: DXA. |
Lower limbs. | Isokinetic dynamometer and DXA are commonly used for assessing leg strength and mass, but are costly and require specialized equipment and training, limiting use in non-clinical settings. |
| Brazil | Neto et al. [56] | 786 adults (66.3 yrs) | Strength-to-mass ratio. 1. Field MQI, highest reading divided by the subject’s body mass index. 2. Laboratory MQI, ratio of grip strength to the entire arm muscle in kilograms measured by DXA. |
Test: HGS. Instrument: for muscle strength: isometric dynamometer (HGS), for arm muscle lean mass: DXA (body composition). |
Upper limbs (arm). | Handgrip and DXA assessments are reliable for community use, though DXA's expense and equipment needs can limit broader accessibility. |
| Brazil | Neto et al. [57] | 35 older men (65.8 ± 3.9 yrs) | Strength-to-mass ratio. 1. Laboratory MQI ratio of predicted 1RM leg extension to the entire lower limbs muscle in kilograms measured by DXA. 2. Field MQI ratio of predicted 1RM leg extension divided by the BMI. |
Test: knee extensors. Instrument: for muscle strength: 10 RM test for leg extension, for lean mass: DXA. |
Lower limbs. | These types of dynamometers are used to assess functional muscle quality and feasible for community programmes, while DXA provides a detailed assessment of mass, but is limited to clinical settings due to its cost. |
| Brazil | Nogueira Paranhos Amorim et al. [58] | 222 community-dwelling women | Strength-to-mass ratio. MQ, ratio of grip strength to arm muscle mass (kg/kg). |
Test: HGS. Instrument: for muscle strength: isometric dynamometer (HGS), for arm muscle lean mass: DXA (body composition). |
Upper limbs (arm). | Handgrip and DXA assessments are reliable for community use, though DXA's expense and equipment needs can limit broader accessibility. |
| Portugal | Nunes et al. [59] | (≥ 60 yrs). | Strength-to-mass ratio. MQI, total strength 1RM (the sum of load in kg of 1RM tests in CP, KE, PC)/total body skeletal muscle mass. |
Test: chest press (CP), knee extension (KE) and preacher curl (PC). Instrument: for muscle strength: 1RM test assessed on CP, KE, and PC performed in this exact order, for skeletal muscle mass Kim et al. proposed formula for lean soft tissue values by BIA. |
Upper and lower limbs (together). | These types of dynamometers are used to assess functional muscle quality and feasible for community programmes, BIA is accessible and low-cost for community settings but lacks the precision of imaging techniques. |
| South Korea | Oh et al. [60] | 246 community-dwelling women (≥ 60 yrs). | Strength-to-mass ratio. MQ, ratio of strength to muscle mass. Ratio of isokinetic torque at the knee to lean mass in the leg (Nm/kg). |
Test: knee extensors and extensors. Instrument: for muscle strength: isokinetic dynamometer, for lean mass: DXA. |
Lower limbs. | Isokinetic dynamometer and DXA are commonly used for assessing leg strength and mass, but are costly and require specialized equipment and training, limiting use in non-clinical settings. |
| Japan | Osuka et al. [61] | 64 obese older women (67.05 ± 5.46 yrs) | Functional muscle quality, as muscle strength per muscle mass (muscle thickness). | Test: knee extension. Instrument: isometric knee extensor strength in the dominant leg using a handheld dynamometer, for muscle mass: B-mode ultrasound. |
Lower limbs (quadriceps muscle). | These types of dynamometers are used to assess functional muscle quality and feasible for community programme and ultrasound allows detailed assessment of muscle quality, though high cost and operator expertise limits their community accessibility. |
| USA | Park et al. [62] | 88 obese women (69.4 ± 6.06 yrs) | Strength-to-mass ratio. MQ, muscle strength per regional muscle mass (Nm/kg). Leg specific force/leg muscle mass or arm specific force/arm muscle mass. |
Test: HGS and knee extension. Instrument: for muscle strength: isometric dynamometer (HGS), isokinetic dynamometer for KES, for leg or arm muscle mass: DXA. |
Upper and lower limbs (separately). | Isokinetic dynamometer and DXA are commonly used for assessing leg strength and mass, but are costly and require specialized equipment and training, limiting use in non-clinical settings. |
| Italy | Pancera et al. [63] | 220 octogenarians (≥ 80 yrs) | Strength-to-mass ratio. MQ, of the dominant leg was calculated as the ratio of isometric quadriceps muscle PT to leg lean mass (Nm/kg). |
Test: knee extension. Instrument: for muscle strength: computerized dynamometer, for muscle mass: BIA. |
Lower limbs. | HGS and BIA are accessible and inexpensive, ideal for community and clinical use, but there are more specific imaging techniques. |
| Brazil | Pinto et al. [64] | 66 older women with previous RT experience (68.8 ± 4.6 yrs) | Strength-to-morphology ratio. MQ, force per unit of muscle mass. Calculated by dividing the 1RM knee extension value by the sum of the QF muscle thickness. |
Test: knee extension dominant leg. Instrument: for muscle strength: 1RM test in knee extension, for quadriceps muscle femoris volume: B-mode ultrasound derived muscle thickness. |
Lower limbs (quadriceps muscle). | These types of dynamometers are used to assess functional muscle quality and feasible for community programme and ultrasound allows detailed assessment of muscle quality, though high cost and operator expertise limits their community accessibility. |
| Brazil | Radaelli [65] | 38 older women (≥ 70 yrs) | Strength-to-morphology ratio. MQ, maximal strength produced per unit of muscle mass. Calculated by dividing maximal rate of torque development by muscle quality echo intensity. |
Test: knee extensors. Instrument: for muscle strength: isokinetic dynamometer, for muscle echo intensity: B-mode ultrasound. |
Lower limbs. | Isokinetic dynamometer and DXA are commonly used for assessing leg strength and mass, but are costly and require specialized equipment and training, limiting use in non-clinical settings. |
| Brazil | Radaelli et al. [66] | 156 older women (65-79 yrs) | Strength-to-morphology ratio. MQ, knee extension 1-RM value of the dominant leg was divided by the MT of the quadriceps muscle of the dominant leg. |
Test: knee extension dominant leg. Instrument: for muscle strength: 1RM test in knee extension, for quadriceps muscle femoris volume: B-mode ultrasound derived muscle thickness. |
Lower limbs (quadriceps muscle). | These types of dynamometers are used to assess functional muscle quality and feasible for community programme and ultrasound allows detailed assessment of muscle quality, though high cost and operator expertise limits their community accessibility. |
| Estonia | Rava et al. [67] | 2618 older adults (70-79 yrs) | Strength-to-mass ratio. 1. MQ upper extremity, handgrip strength by arm lean mass (kg/kg). 2. MQ lower extremity, strength of isometric quadriceps muscle by leg lean mass (kg/kg). |
Test: HGS and isometric leg extension. Instrument: for muscle strength: isometric dynamometer (HGS), custom isometric dynamometer for leg strength, for muscle mass assessment: DXA. |
Upper and lower limbs (separately). | These types of dynamometers are used to assess functional muscle quality and feasible for community programme and DXA commonly applied muscle quality data; the expense and equipment needs of DXA may limit its community use. |
| Iceland | Reinders et al. [68] | 30 stable inpatients (68 ± 8 yrs) | Strength-to-morphology ratio. MQ, the ratio of muscle strength and muscle area. |
Test: knee extension dominant leg. Instrument: for muscle strength: isokinetic dynamometer, 1RM test in knee extension, for leg muscle area CT. |
Lower limbs. | Isokinetic dynamometry and CT offer detailed assessments but are expensive and less feasible for broad application. |
| Portugal | Ribeiro et al. [69] | 36 sedentary elderly women (66.0 ± 8 yrs) | Strength-to-mass ratio. 1. MQI: 1RM load from the chest press (CP), knee extension (KE), and preacher curl (PC) by fat-free mass (FFM). 2. Upper limb MQ: 1RM of the PC by arm lean mass. 3. Lower limb MQ: 1RM of the KE by leg lean mass. |
Test: chest press (CP), knee extension (KE) and preacher curl (PC). Instrument: for muscle strength: 1RM test assessed on CP, KE, and PC performed in this exact order, for fat free mass: DXA. |
Upper and lower limbs (together and separately). | These types of dynamometers are used to assess functional muscle quality and feasible for community programmes, DXA provide commonly applied muscle quality data; the expense and equipment needs of DXA may limit its community use. |
| Brazil | Pinto et al. [64] | 66 older women with previous RT experience (68.8 ± 4.6 yrs) | Strength-to-morphology ratio. MQ, force per unit of muscle mass. Calculated by dividing the 1RM knee extension value by the sum of the QF muscle thickness. |
Test: knee extension dominant leg. Instrument: for muscle strength: 1RM test in knee extension, for quadriceps muscle femoris volume: B-mode ultrasound derived muscle thickness. |
Lower limbs (quadriceps muscle). | These types of dynamometers are used to assess functional muscle quality and feasible for community programme and ultrasound allows detailed assessment of muscle quality, though high cost and operator expertise limits their community accessibility. |
| Brazil | Radaelli [65] | 38 older women (≥ 70 yrs) | Strength-to-morphology ratio. MQ, maximal strength produced per unit of muscle mass. Calculated by dividing maximal rate of torque development by muscle quality echo intensity. |
Test: knee extensors. Instrument: for muscle strength: isokinetic dynamometer, for muscle echo intensity: B-mode ultrasound. |
Lower limbs. | Isokinetic dynamometer and DXA are commonly used for assessing leg strength and mass, but are costly and require specialized equipment and training, limiting use in non-clinical settings. |
| Brazil | Radaelli et al. [66] | 156 older women (65-79 yrs) | Strength-to-morphology ratio. MQ, knee extension 1-RM value of the dominant leg was divided by the MT of the quadriceps muscle of the dominant leg. |
Test: knee extension dominant leg. Instrument: for muscle strength: 1RM test in knee extension, for quadriceps muscle femoris volume: B-mode ultrasound derived muscle thickness. |
Lower limbs (quadriceps muscle). | These types of dynamometers are used to assess functional muscle quality and feasible for community programme and ultrasound allows detailed assessment of muscle quality, though high cost and operator expertise limits their community accessibility. |
| Estonia | Rava et al. [67] | 2618 older adults (70-79 yrs) | Strength-to-mass ratio. 1. MQ upper extremity, handgrip strength by arm lean mass (kg/kg). 2. MQ lower extremity, strength of isometric quadriceps muscle by leg lean mass (kg/kg). |
Test: HGS and isometric leg extension. Instrument: for muscle strength: isometric dynamometer (HGS), custom isometric dynamometer for leg strength, for muscle mass assessment: DXA. |
Upper and lower limbs (separately). | These types of dynamometers are used to assess functional muscle quality and feasible for community programme and DXA commonly applied muscle quality data; the expense and equipment needs of DXA may limit its community use. |
| Iceland | Reinders et al. [68] | 30 stable inpatients (68 ± 8 yrs) | Strength-to-morphology ratio. MQ, the ratio of muscle strength and muscle area. |
Test: knee extension dominant leg. Instrument: for muscle strength: isokinetic dynamometer, 1RM test in knee extension, for leg muscle area CT. |
Lower limbs. | Isokinetic dynamometry and CT offer detailed assessments but are expensive and less feasible for broad application. |
| Portugal | Ribeiro et al. [69] | 36 sedentary elderly women (66.0 ± 8 yrs) | Strength-to-mass ratio. 1. MQI: 1RM load from the chest press (CP), knee extension (KE), and preacher curl (PC) by fat-free mass (FFM). 2. Upper limb MQ: 1RM of the PC by arm lean mass. 3. Lower limb MQ: 1RM of the KE by leg lean mass. |
Test: chest press (CP), knee extension (KE) and preacher curl (PC). Instrument: for muscle strength: 1RM test assessed on CP, KE, and PC performed in this exact order, for fat free mass: DXA. |
Upper and lower limbs (together and separately). | These types of dynamometers are used to assess functional muscle quality and feasible for community programmes, DXA provide commonly applied muscle quality data; the expense and equipment needs of DXA may limit its community use. |
| Brazil | Ribeiro et al. [70] | 26 adult women (64.8 ± 3.2 yrs) | Strength-to-mass ratio. 1. MQI: 1RM load from the chest press (CP), knee extension (KE), and preacher curl (PC) by fat-free mass (FFM). 2. Upper limb MQ: 1RM of the PC by arm lean mass. 3. Lower limb MQ: 1RM of the KE by leg lean mass. |
Test: chest press (CP), knee extension (KE) and preacher curl (PC). Instrument: for muscle strength: 1RM test, assessed on CP, KE, and PC performed in this exact order, for fat free mass: DXA. |
Upper and lower limbs (together and separately). | These types of dynamometers are used to assess functional muscle quality and feasible for community programmes, DXA provide commonly applied muscle quality data; the expense and equipment needs of DXA may limit its community use. |
| Italy | Rondanelli et al. [71] | 27 postmenopausal and normoweight older women (67.6 ± 5.1 yrs) | Strength-to-mass ratio. MQ, strength/muscle mass. |
Test: HGS and isometric leg extension. Instrument: for muscle strength: isometric dynamometer (HGS), isometric dynamometer for leg extension test, for muscle mass assessment: DXA. |
Upper limbs (for both arms). | These types of dynamometers are used to assess functional muscle quality and feasible for community programmes and DXA provide commonly applied muscle quality data; the expense and equipment needs of DXA may limit its community use. |
| USA | Salas-Groves et al. [72] | 32 older women (≥ 65 yrs) | Strength-to-mass ratio. MQ, the ratio of muscle strength to lean body mass (kg/kg). |
Test: HGS. Instrument: for muscle strength: camry digital hand dynamometer (HGS), for muscle mass and body composition: DXA. |
Upper limb. | Handgrip and DXA assessments are reliable for community use, though DXA's expense and equipment needs can limit broader accessibility. |
| USA | Schroeder et al. [73] | 2725 older adults (74.8 ± 4.7 yrs) | Strength-to-mass ratio. MQ, maximal composite strength score in units of kg divided by DXA total lean mass in units of kg (kg/kg). |
Test: bilateral leg press, leg extension, leg flexion, latissimus pull-down, and chest press. Instrument: for muscle strength: 1RM test in LP, LE, LF, LPD and CP, for lean mass: DXA. |
Lower limbs. | These types of dynamometers are used to assess functional muscle quality and feasible for community programmes and DXA provide commonly applied muscle quality data; the expense and equipment needs of DXA may limit its community use. |
| South Korea | Seo et al. [74] | 34 older women | Strength-to-morphology ratio. MQ, maximal voluntary contraction per muscle mass (cross sectional area). |
Test: knee extension dominant leg. Instrument: for muscle strength: isokinetic dynamometer, 1RM test in knee extension, for leg muscle area: CT. |
Lower limbs. | Isokinetic dynamometry and CT offer detailed assessments but are expensive and less feasible for broad application. |
| USA | Shin et al. [75] | 25 physically independent older women (67.6 ± 5.1 yrs) | Strength-to-mass ratio. MQ, muscle strength (maximal voluntary contraction)/mineral-free lean mass (MFLM). 1. Upper legs MQ: knee extension MVC + knee flexion MVC/ MFLM upper legs. 2. Ankle MQ: MVC of ankle dorsiflexion and plantarflexion/ MFLM lower leg. Strength of both legs was summed and normalized to the combined MFLM to calculate MQ. |
Test: knee extension, knee flexion, ankle dorsiflexion and ankle plantarflexion. Instrument: for muscle strength: isokinetic dynamometer for knee extension, knee flexion, ankle dorsiflexion and ankle plantarflexion test, for lean mass: DXA. |
Lower limbs. | Isokinetic dynamometer and DXA are commonly used for assessing leg strength and mass, but are costly and require specialized equipment and training, limiting use in non-clinical settings. |
| Brazil | Silva Junior et al. [76] | 159 older adults admitted in physical medicine and rehabilitation division (80.3 yrs, 107 females) | Strength-to-mass ratio. MQI, muscle strength/ muscle mass. Upper body: 1. MQI field: HGS/BMI. 2. MQI lab: HGS/arm lean mass (DXA). 3. MQI lab: 10RM leg extension/lean mass from both legs (DXA). |
Test: HGS and leg extension. Instrument: for muscle strength: isometric dynamometer (HGS) and 10 RM obtained from the leg extension exercise, for muscle and lean mass assessment: DXA. |
Upper and lower limbs (separately). | These types of dynamometers are used to assess functional muscle quality and feasible for community programmes and DXA provide muscle quality data; the expense and equipment needs of DXA may limit its community use. |
| Germany | Spira et al. [77] | 47 older adults (mean 70 yrs) | Strength-to-mass ratio. MQ, as hand grip strength related to arm muscle mass. |
Test: HGS. Instrument: for muscle strength: Smedley Hand Dynamometer (HGS), for muscle mass and body composition: DXA. |
Upper limb. | Handgrip and DXA assessments are reliable for community use, though DXA's expense and equipment needs can limit broader accessibility. |
| Germany | Stotz et al. [78] | 88 older adults (≥65 yrs) | Strength-to-mass ratio. Trunk muscle quality as strength relative to lean mass. Absolute trunk flexion and extension torque/lean tissue mass. |
Test: trunk flexion and extension. Instrument: for muscle strength: isokinetic concentric trunk flexion and extension dynamometer, for lean tissue mass: inbody (BIA). |
Trunk. | Isokinetic devices are reliable for targeted measures but costly for routine community use. BIA is accessible and low-cost, ideal for community and clinical use, but lack detailed assessment capability. |
| USA | Straight et al. [79] | 59 older women | Strength-to-mass ratio. MQ, leg extension power to lower body mineral free lean mass (W/kg). |
Test: knee extension. Instrument: for muscle power: nottingham power rig, for lean mass: DXA. |
Lower limbs. | DXA is accurate for muscle mass assessment, but high-tech equipment of both limits accessibility. |
| Australia | Sui et al. [80] | (≥ 65 yrs) | Strength-to-mass ratio. MQ, expressed as HGS in relation to arm lean mass. |
Test: HGS. Instrument: for muscle strength: isometric dynamometer (HGS), for muscle mass and body composition: DXA. |
Upper limb. | Handgrip and DXA assessments are reliable for community use, though DXA's expense and equipment needs can limit broader accessibility. |
| USA | Tracy et al. [81] | 72 older adults (69.5 ± 6.1 yrs) | Strength-to-morphology ratio. MQ, strength per unit of muscle mass (muscle volume) (kg/cm3). Isometric and 1-RM strength values (N and kg, respectively)/ muscle volume. |
Test: knee extension. Instrument: for muscle strength: 1RM strength test, isometric force production, and isokinetic peak torque were assessed in the knee extensors, for muscle mass and volume: MRI. |
Lower limbs (quadriceps muscle). | Isokinetic devices are reliable for targeted measures but costly. MRI is effective but costly and complex for community implementation. |
| Brazil | Vilaca et al. [82] | 39 older women (69.0 ± 6.2 yrs) | Strength-to-mass ratio. MQ, muscle strength to lean mass ratio. 1. Upper limb: HGS/ lean mass of the dominant upper limb (kg/kg). 2. Lower limb: bilateral KES (lean mass of the lower limbs (kg/kg). |
Test: HGS and knee extension. Instrument: for muscle strength portable hydraulic dynamometer, 1RM knee extension test, for muscle mass and body composition: DXA. |
Upper and lower limbs. | These types of dynamometers are used to assess functional muscle quality and feasible for community programmes and DXA allow assessment of muscle quality data; the expense and equipment needs of DXA may limit its community use. |
| Brazil | Vilaca et al. [83] | 838 older adults (60-82 yrs) | Strength-to-mass ratio. MQ, muscle strength to lean mass ratio. 1. Upper limb: HGS/ lean mass of the dominant upper limb (kg/kg). 2. Lower limb: bilateral KES (lean mass of the lower limbs (kg/kg). |
Test: HGS and knee extension. Instrument: for muscle strength portable hydraulic dynamometer, 1RM knee extension test, for muscle mass and body composition: DXA. |
Upper and lower limbs. | These types of dynamometers are used to assess functional muscle quality and feasible for community programmes and DXA provide assessment of muscle quality data; the expense and equipment needs of DXA may limit its community use. |
| Spain | Virto et al. [84] | 74 older adults (78.1 ± 5.8 yrs, 38 men) | Strength-to-mass ratio. MQI, defined as handgrip strength (kg) divided by relative skeletal muscle mass (kg). |
Test: HGS. Instrument: Camry EH101 electronic handheld dynamometer (HGS), for muscle skeletal muscle mass: BIA. |
Upper and lower limbs. | HGS and BIA are accessible and inexpensive, ideal for community and clinical use, but there are more specific imaging techniques. |
| Brazil | Vojciechowski et al. [85] | 96 community-dwelling older women (73.9 ± 5.6 yrs) | Strength-to-morphology ratio. MQ, quadriceps muscle peak torque to cross-sectional area (CSA). |
Test: knee extension, knee flexion, ankle dorsiflexion and ankle plantarflexion. Instrument: for muscle strength, isokinetic dynamometer for knee extension, knee flexion, ankle dorsiflexion and ankle plantarflexion test, for cross sectional area: MRI. |
Lower limb (quadriceps muscle). | Isokinetic devices are reliable for targeted measures but costly. MRI is effective but costly and complex for community implementation. |
| Italy | Volpato et al. [86] | 342 older men (60-96 yrs) | Strength-to-morphology ratio. MQ, ratio of ankle strength (dorsiflexion + plantarflexion) per muscle area (kg/cm²). |
Test: ankle dorsiflexion and plantar flexion. Instrument: for muscle strength handheld dynamometer, for muscle cross sectional area measured at 66% of the tibial length starting from the tibiotarsal joint peripheral, quantitative CT. |
Lower limbs (ankle, plantar). | These types of dynamometers are used to assess functional muscle quality and feasible for community programmes, pQCT provides detailed density analysis but is expensive and less accessible for community use. |
| UK | Walsh et al. [87] | 23 older adults (65-75 yrs) | Strength-to-mass ratio. 1. Upper leg MQ, sum of knee flexor and extensor peak torque/ upper leg muscle mass. 2. Lower leg MQ, sum of ankle plantarflexion and dorsiflexion maximum torque divided by lower leg muscle mass. 3. Leg total MQ, sum of all strength measures/ sum of upper and lower leg muscle mass (Nm/kg). |
Test: knee flexors and extensors test, ankle plantarflexion and dorsiflexion test. Instrument: for muscle strength isokinetic dynamometer, for muscle mass: DXA. |
Lower limbs. | Isokinetic dynamometer and DXA are commonly used for assessing leg strength and mass, but are costly and require specialized equipment and training, limiting use in non-clinical settings. |
| Japan | Yamada et al. [88] | 75 older women (65-80 yrs) | Strength-to-morphology ratio. MQ, knee extension torque divided by the quadriceps muscle femoris muscle thickness (Nm/cm). |
Test: knee extension. Instrument: for muscle strength: isokinetic dynamometer for knee extension, for quadriceps muscle femoris volume: B-mode ultrasound derived muscle thickness. |
Lower limbs (quadriceps muscle). | Isokinetic dynamometer and ultrasound allow assessment of muscle quality, though high cost limits and operator expertise their community accessibility. |
| Japan & Taiwan | Yamaguchi et al. [89] | 77 active older women (65-80 yrs) | Strength-to-mass ratio. MQ, grip strength normalized to skeletal muscle mass (kg/kg). |
Test: HGS. Instrument: for muscle strength: isometric dynamometer (HGS), for muscle mass assessment: BIA. |
Upper limbs. | HGS and BIA are accessible and inexpensive, ideal for community and clinical use, but there are more specific imaging techniques. |
| South Korea | Yoon et al. [90] | 1253 older adults (78.1 ± 5.8 yrs) | Strength-to-mass ratio. MQ, the ratio of the strength measured to the entire corresponding leg muscle mass in kg measured by DXA. |
Test: knee extension. Instrument: for muscle strength: isokinetic dynamometer, for muscle mass: DXA. |
Lower limbs. | Isokinetic dynamometer and DXA are commonly used for assessing leg strength and mass, but are costly and require specialized equipment and training, limiting use in non-clinical settings. |
| China | Yu et al. [92] | 46 older women (69 ± 4 yrs) | Strength-to-mass ratio. MQ, grip strength normalized to skeletal muscle mass (kg/kg). |
Test: HGS. Instrument: for muscle strength: isometric dynamometer (HGS), for muscle mass assessment: BIA. |
Upper limbs. | HGS and BIA are accessible and inexpensive, ideal for community and clinical use, but there are more specific imaging techniques. |
The literature shows considerable heterogeneity in the operational definition of MQ (Table 2). However, most definitions within the functional domain align with indirect measures of muscle function relative to muscle mass, typically expressed as strength per unit of muscle mass, a definition employed by 70 studies. Various methods are employed to assess muscle strength, including PT, 1RM, 5RM, isometric strength, and others. Muscle mass is generally quantified through metrics such as body mass, muscle thickness, muscle volume, cross-sectional area, and lean mass. Among the alternative definitions, four studies utilize established formulas to measure MQ functionally. One of these applies the muscle quality index (MQI) formula: MQI = [(L - 0.5) × body mass × g × 5] / Tsit-tostand. The remaining three studies use the formula MQI = [(leg length × 0.4) × body mass × gravity × 10] / time sitto-stand. Additionally, two studies employ the power-tomass ratio, while another uses the specific tension-to-mass ratio as a measure of MQ.
The review identified various non-invasive tools for assessing functional MQ in older adults, focusing on strength and mass (Table 2). For strength, 16 studies used handheld dynamometers for handgrip strength (HGS), knee extension involved 23 isokinetic, 6 handheld isometric, and 1 combined method. Leg power was assessed in 4 studies, RM in 10, and 11 combined upper and lower strength. For muscle mass, 44 studies used DXA, 11 ultrasound, 12 BIA, 4 MRI, and 8 CT, with CT and ultrasound assessing CSA, volume, or composition.
The most effective non-invasive tools for assessing functional MQ in older adults balance accuracy and accessibility (Table 3). The MQI is emerging to assess strength– mass relationships. BIA use has increased, with 92% of studies published since 2020, and ultrasound use has grown since 2012, especially with portable devices enhancing accessibility. However, heterogeneity and lack of consensus in definitions limit comparability between studies, with specific tool limitations detailed in Table 3.
Table 3.
carcinoma.
| Tool | Advantage | Disadvantage | Time and portability | Cost and training |
|---|---|---|---|---|
| Quantification of muscle strength | ||||
| Isometric dynamometry (hand-held dynamometers for upper and and lower limbs) [2, 31] | - High reliability and validity for indirect measurement of muscle strength. - Easy to use and accessible for community studies. |
- It does not directly measure muscle mass, only strength. - It only measures isometric strength lacking dynamic muscle contraction, less relevant than isokinetic. |
Fast measurement and high portability. | Low cost and training. |
| Isokinetic dynamometry [11, 43, 92] | - High accuracy in measuring muscle force and torque at different speeds. - High validity and reliability to assess specific force (isokinetic, isotonic and isometric). - More physiologically relevant than isometric as it involves dynamic muscle contractions. |
- Expensive equipment and requires trained operators. - It is not portable, which limits its accessibility in community settings. |
Moderate measurement and low portability. | Medium-high cost and training. |
| RM [95] | - Accuracy to determine the maximum force in a specific exercise. - Inexpensive and accessible, does not require advanced equipment (can be performed in any environment with free weights or machines). |
- May be a risk of injury, especially in older people or those with health conditions. - Specialised equipment, expensive and requires training. |
Moderate measurement and high portability. | Medium cost and training. |
| Quantification of muscle mass | ||||
| BIA [9, 70, 100] | - Non-invasive, quick, and accessible technique. - Uses whole-body electrical conductivity to estimate body composition. |
- Lower accuracy compared to tools such as DXA or MRI. - Sensitive to external variables (hydration, temperature). |
Fast measurement and medium portability. | Medium cost and low training. |
| DXA [2, 70, 89, 97, 98] | - High validity and accuracy for measuring lean mass, appedicular skeletal muscle mass, fat and bone mineral density. - Whole body and body segment. - Good indicator of the relationship between strength and muscle mass. - Low-radiation technique. |
- High cost, need for specialised equipment and operators. - Non portable. - Cannot evaluate an individual muscle or assess muscle quality. |
Moderate measurement and low portability. | High cost and specialised operators. |
| B mode Ultrasound [9, 98, 99] | - It measures muscle architecture (CSA, EI, FL, PA) as an accurate marker of muscle quality. - Good precision. - It is non-invasive, no radiation, fast and can be portable. |
- Measurements highly dependent on operator expertise. - Intermachine and interoperator variability. - Intermediate cost and technical training requirement. |
Fast - moderate measurement and high to medium portability. | Medium cost and specialised operators. |
| CT [9, 12, 13, 97, 98] | - Excellent accuracy and detail for measuring muscle composition, in this case muscle cross-sectional area. - High validity in the assessment of muscle quality. - Quantitative and qualitative assessment. |
- High cost and radiation exposure. - Not routinely applicable in community studies. - Non portable. |
Extended measurement and low portability. | Very high cost and specialised operators. |
| MRI [9, 12, 13, 97] | - The most accurate tool for measuring body composition (muscle volume and quality). - No radiation exposure. - Very high validity. - Quantitative and qualitative assessment. |
- Very costly and not accessible for most community studies. - Operational complexity, non portable equipment and training requirements. |
Extended measurement and low portability. | Very high cost and specialised operators. |
Discussion
This scoping review aimed to assess studies on non-invasive tools for evaluating functional MQ in older adults, focusing on both effectiveness and practical aspects. Objectives included identifying commonly used methods, examining recent advances, and exploring limitations and practical factors, such as cost, accessibility, and training, that influence tool selection and the feasibility of community-based applications.
MQ is a central concept in the study of sarcopenia and other age-related musculoskeletal disorders, as it appears to be more sensitive to age-associated changes than either strength or muscle mass, thus serving as a valuable indicator of muscle health and function [3, 7]. However, although the findings of this review demonstrate substantial heterogeneity in the definitions of MQ, definitions of functional MQ primarily involve indirect assessments of muscle performance relative to muscle mass, typically expressed as strength per unit of muscle mass, consistent with findings from the literature [3, 7]. This definition, adopted by 70 studies, is grounded in the same fundamental concept of the strength-to-mass ratio; nonetheless, heterogeneity arises due to the variability in the methods used to assess both strength and muscle mass. Consequently, despite the use of a common conceptual framework for MQ, variations in measurement techniques result in inconsistencies in the outcomes obtained, underscoring the need for methodological standardisation to enhance the comparability across studies.
The diversity of tools used to measure muscle strength and mass contributes to heterogeneity in functional MQ assessments. Among non-invasive methods, 16 studies measured HGS with handheld dynamometers; for knee extension, 23 used isokinetic devices, 6 handheld isometric, and 1 both. Additionally, 4 assessed leg power, 10 used RM, and 11 combined measures.
An additional source of heterogeneity may stem from differences in the anatomical sites used to evaluate muscle strength and mass. Twelve studies have assessed strength in a specific muscle group, such as the knee extensors or handgrip, while estimating muscle mass from whole-body or appendicular lean mass derived from DXA or BIA. This discrepancy between the anatomical sites used for strength and muscle mass assessment, may not accurately represent the functional relationship between muscle size and performance, which may contribute to the observed variability in MQ indices across studies [5, 9].
Evaluating muscle force output is a critical clinical consideration, and one of the most reliable methods for objectively measuring strength is through the use of dynamometry. Dynamometry quantifies and provides objective measurements of force, torque, or power generated during muscle contraction and is classified as the primary laboratory-based methodology. There are several types of dynamometry, each addressing different assessment needs. Among the types identified in the literature are isokinetic dynamometry and handheld dynamometry (isometric). As for the gold standard, isokinetic dynamometry is considered the most valid and reliable method for measuring muscle force. These computerised machines are capable of providing multiple metrics related to muscle force, allowing accurate force measurements at various angles and speeds, while minimising problems associated with the measurements [11, 92, 93]. However, isokinetic dynamometers are expensive and not portable, thereby making them unsuitable for routine clinical examinations in many settings [11, 93]. On the other hand, isometric dynamometers, such as hand-held dynamometers (HHD), are more accessible in clinical practice due to their portability, simplicity, and lower cost compared to isokinetic devices. However, the information they provide is more limited, as they primarily offer a portable, quantitative method for assessing isometric muscle strength and power [15, 92]. The affordability of these dynamometers may justify their widespread use in clinical settings, but although correlations between HHD and isokinetic measurements exist and they are considered reliable and valid, they do not offer the same level of detailed information as isokinetic testing [2, 41, 94].
On the other hand, among the most commonly used field tests for measuring strength is the one repetition maximum (1RM) strength test, defined as the maximum weight that can be lifted once with correct lifting technique. This test is widely used to determine the maximum force for a specific movement, is simple, safe, inexpensive and is considered the gold standard in non-laboratory situations. Furthermore, it follows the same patterns as those performed by individuals during their regular training sessions or in daily life allowing the assessment of strength in multi-joint exercises [10, 95].
Power assessment is crucial for evaluating an individual’s ability to generate force rapidly, which is the product of force and velocity [10]. This can be measured using isokinetic dynamometers or more specific methods, such as the Nottingham Power Rig and the PLEE power lift, which are the ones used in the studies analizes in this review. Both tools exhibit high precision in measuring explosive power, integrating force and velocity. In terms of more specific differences, the Nottingham Power Rig is particularly suited for clinical populations or isolated leg tests, whereas the PLEE power lift is primarily used for assessing athletic performance in functional movements being more challenging to perform safely in older populations [53, 96].
Muscle mass evaluation can be assessed using a range of non-invasive methods, among the methods used in the studies analysed by the review, DXA was employed in 44 studies, ultrasound was used in 11 studies, BIA was utilized in 12 studies, MRI in 4 studies, and CT in 8 studies. CT and MRI are considered the gold standard for assessing muscle composition being endorsed methodologies by EWGSOP2, especially MRI. These methods allow both qualitative and quantitative assessments of body composition, and are notable for their high ability to differentiate tissue types, such as intramuscular fat infiltration, muscle volume, cross-sectional area and other structural characteristics [12, 13]. While CT provides a faster and more cost-efficient option, it is associated with radiation exposure, whereas MRI offers a radiation-free alternative, albeit at a higher cost and requiring greater technical expertise. Despite their strong correlation in clinical evaluations of muscle quantity and quality, their high cost, specialized equipment requirements, and the need for trained personnel limit their use in routine community-based practice [12, 13].
Another commonly utilized method for evaluating muscle composition is DXA, which is the most frequently employed tool in clinical practice for assessing body composition and diagnosing sarcopenia [2, 97]. This is evidenced by the findings of the conducted review, where 56% of all analyzed studies utilized DXA to measure muscle mass. However, there is considerable controversy in the literature regarding the appropriateness of referring to DXA as the “gold standard”. On the one hand, DXA is considered a precise, valid, reproducible, and widely available imaging modality that quantifies lean mass, appendicular skeletal muscle mass, fat, and bone mineral density, and it is also a good indicator of the relationship between muscle strength and muscle mass. However, it does not measure muscle composition at a more structural level, can be influenced by hydration status, is relatively expensive, and is not portable [2, 97].
Ultrasonography is increasingly recognized as a fast, noninvasive, and accessible method for musculoskeletal assessment, enabling the evaluation of both quantitative and qualitative muscle parameters [9, 98, 99]. Recent systematic reviews assessing the validity and reliability of ultrasonography for skeletal muscle evaluation have reported high interclass correlation coefficients and confirmed its comparability with other imaging modalities [2, 9, 98, 99]. Despite ongoing efforts to standardize these measurements, their high operator dependency underscores the need for further research to enhance their clinical applicability and to increase lack of reference [9, 98, 99]. Notably, recent studies have begun to demonstrate strong correlations between ultrasound-derived parameters and other reference measures of muscle mass [9, 98, 99].
BIA is an effective non-imaging method for estimating total or appendicular skeletal muscle mass by measuring the body’s electrical conductivity and applying predictive equations. While it is non-invasive, quick, and accessible, its accuracy depends significantly on the predictive equation used and can be influenced by factors such as hydration status [9, 100]. Compared to reference methods like DXA and CT, BIA may slightly overestimate or underestimate skeletal muscle mass, but it generally demonstrates strong correlations with these techniques [2, 70].
A major challenge of these tools is the lack of standardised assessment criteria, makes it difficult to identify the most appropriate and consistently applied methods across studies. The most efficient tools for assessing the functional domain of MQ aim to capture muscle function (strength, power) while considering structural properties (mass). They must be reliable, reproducible, and capable of providing objective, clinically meaningful data. CT, MRI, and DXA are the most commonly used for structural and compositional assessment, with MRI and DXA being preferable due to the absence of radiation and greater accessibility. However, ultrasound and BIA offer practical, low-cost, and rapid alternatives for non-invasive assessment, despite slightly lower accuracy [2, 9, 12, 98]. In the context of strength, isokinetic dynamometry, 1RM test and hand-held isometric dynamometers assess muscle strength, but vary in accuracy, cost and applicability. Isokinetic dynamometry offers the greatest accuracy, but is limited by cost and portability. The 1RM test is inexpensive and practical, but lacks the detailed measurements of isokinetic devices. Portable dynamometers are more accessible and affordable, but provide less detailed data [10, 11, 94]. Each technique relies on different technologies and focuses on specific dimensions of functional MQ, underscoring the importance of careful selection based on the goals of the assessment and available resources. This can be seen in detail in Table 3, which summarises the advantages and limitations of each tool.
Current non-invasive tools for assessing the functional domain of MQ in older adults are limited primarily by the absence of standardized criteria and significant methodological heterogeneity. This variability hinders direct comparisons across studies and complicates the identification of the most effective tool for this purpose. Notably, other papers and reviews on MQ have similarly reported challenges stemming from the heterogeneity of assessment methods [3, 4, 9]. A detailed evaluation of the strengths and weaknesses of each method, as outlined in Table 3, highlights the specific limitations inherent to each approach.
When interpreting Table 3, the selection of a MQ assessment tool should consider the specific context in which it will be applied. For community screening or large epidemiological studies, portable and low-cost tools such as hand-held dynamometry or BIA may be more feasible, despite their lower precision. In routine clinical settings, methods that balance feasibility and accuracy, such as one-repetition maximum testing or regional assessments with DXA or ultrasound, may provide more informative estimates of functional MQ. Conversely, research settings requiring high precision may benefit from techniques such as isokinetic dynamometry combined with imaging methods like MRI or CT [2, 9].
The EWGSOP stresses the importance of assessing both muscle quality and quantity [2]. However, our review highlights that a clear criterion for selecting a specific assessment tool based on an individual’s characteristics has not yet been established [2, 9]. When assessing the functional domain of MQ, several factors influence the choice of non-invasive tools. These include cost, accessibility and the level of training required to use the tool effectively. In clinical settings, accurate and validated tools such as DXA and MRI are frequently used despite their high cost, limited accessibility and the need for specialised staff. While these direct methods provide reliable data, their limited accessibility creates barriers for widespread use, especially in non-clinical settings [2, 9]. As a result, indirect measures that rely on simpler, portable and more affordable tools, such as BIA or ultrasound are gaining traction in everyday use. However, they are limited by their moderate to low level of precision, compared with those referred above [99]. The increasing adoption of portable ultrasound and BIA devices in community settings highlights the potential for integration of affordable and simple methods in these settings [2]. These approaches offer a practical solution to bridge the gap between clinical and community settings, making MQ assessment more accessible and scalable, especially when resources are limited. While more expensive methods are often reserved for clinical use, simpler and more cost-effective tools are more viable for community settings, where ease of use and affordability are critical for widespread implementation [2, 97, 98]. The need for accessible tools is important because changes in MQ often occur earlier than changes in other functional variables, and detecting these changes might serve as a more sensitive indicator of muscle function preventing further deterioration in health [3, 7]. Key parameters for the selection of MQ assessment methods are summarized in Figure 2.
Figure 2. Key parameters for the selection of muscle quality assessment methods. Conceptual flowchart illustrating how the selection of noninvasive muscle quality assessment tools can be guided by the assessment purpose (screening, monitoring, or research), the target population or setting (community, clinical, or specialised environments), and the available resources (accessibility, equipment, and trained personnel), across morphological and functional domains.
It is essential to expand the evaluation of this parameter to the community level since variations in MQ frequently appear before changes in other functional variables [3, 7]. This encompasses a broad range of uses, from community events and health promotion initiatives to incorporation into exercise and training facilities tailored to the needs of senior citizens. As previously said, accessibility and measurement speed are crucial, particularly in situations with a large number of people and few resources. In this regard, creating and utilizing instruments that are quick, precise, and profitable might be a crucial remedy [3]. A viable substitute are functional quality measurement formulas derived from indirect evaluations of muscle performance in relation to muscle mass [9]. These formulations could be successfully integrated into community settings when muscle examinations are carried out with easily available equipment, allowing for widespread tracking and enhancement of muscle health. Handheld dynamometers, which may be used for both upper and lower limbs and include 1RM evaluations, are among the most widely used and reasonably priced instruments for measuring muscle strength. US and BIA are the most widely available methods for measuring muscle mass in community settings, with BIA being the most straightforward and useful alternative for broad use.
In recent years, several emerging trends have highlighted the growing use of accessible and portable technologies for assessing MQ in community and clinical settings, including handheld dynamometers, portable force measurement devices, and BIA. At the same time, increasing attention has been given to improving the standardization of portable ultrasound based measurements, particularly regarding acquisition protocols and operator training, to enhance reproducibility across studies. In addition, functional indicators such as the MQI have gained interest for evaluating the relationship between muscle strength and muscle mass using simple field-based approaches.
Conclusions
This review highlights the considerable heterogeneity of definitions of MQ in the functional domain, where indirect measures of muscle function in relation to muscle mass predominate. Although the concept itself is based on a shared fundamental framework, relating strength to muscle mass, variability arises primarily from the various methods used to assess both strength and muscle mass. These differences in measurement techniques lead to poorly comparable results between studies, underlining the need for methodological standardisation.
A wide array of non-invasive tools is available for assessing the functional domain of MQ in older adults, with a growing preference for accessible technologies suitable for both clinical and community settings. Among strength measurement tools, handheld dynamometers and 1RM tests stand out as the most straightforward and practical options, particularly for community applications. In contrast, isokinetic dynamometers and power assessments, while offering greater precision, are more specialized and less feasible for widespread use outside clinical or research environments. For muscle mass evaluation, gold-standard tools such as MRI, DXA, and CT provide exceptional accuracy and validity but are often limited by their high cost and complexity, making them less practical for community implementation. More accessible alternatives like BIA and US offer scalable and cost-effective solutions, making them well-suited for use in communitybased settings.
One of the main outcomes of this review is to provide practical recommendations for the selection and use of functional MQ assessment tools, adapted to budgetary constraints and community practice settings. One recommendation involves using formulas to assess functional MQ based on indirect evaluations of muscle performance relative to muscle mass, offering a practical and scalable approach. When combined with accessible tools such as handheld dynamometers, BIA, or ultrasound, these methods can facilitate large-scale monitoring of muscle health in community settings. Another suggestion is to align the anatomical sites for assessing muscle strength and muscle mass; if strength is evaluated at a specific site, muscle mass should also be measured at the same location to ensure consistency and accuracy.
This review has several limitations. Firstly, studies that did not explicitly mention MQ in their title or abstract, or those that defined functional MQ differently from preestablished definitions, may have been excluded. In addition, the scope of the review was limited to individuals aged 60 years and older, potentially overlooking interventions targeting other populations that might offer broader perspectives. However, this approach aligns with the goal of understanding MQ in the ageing population. Finally, the lack of standardisation and the high heterogeneity of tools used to assess MQ justify the decision to conduct a scoping review rather than a systematic review, as it allows for a more flexible exploration of emerging techniques and tools in this area.
A valuable avenue for future research lies in creating personalized protocols for selecting measurement tools, customized to the unique circumstances of each patient.
Declarations
Author contributions
NV conceived the idea and design for the article. NV and XR performed the literature search. NV performed data acquisition, analysis, and/or interpretation. NV prepared the first draft. NV, XR, AM-Z and BG-Z drafted and/or critically revised the work. All authors have read, and approved the final version of the manuscript.
Availability of data and materials
The systematic search queries and data charting methods employed in this study are available upon request. Additionally, our systematic review was registered on the Open Science Framework (OSF) platform on November 4, 2024 with the registration doi (DOI: https://doi.org/10.17605/OSF. IO/QV28T). The authors are committed to fostering transparency in research, and for any inquiries or requests for specific methodological details, we welcome communication with the corresponding author.
Financial support and sponsorship
N.V received a University of Deusto grant from the researcher education program (Award number: FPI UD_2022_10).
Conflicts of interest
Not applicable.
References
1. Clark B, & Manini T. Sarcopenia =/= dynapenia. J Gerontol A Biol Sci Med Sci, 2008, 63(8): 829-834. [Crossref]
2. Cruz-Jentoft A, Bahat G, Bauer J, Boirie Y, Bruyère O, Cederholm T, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing, 2019, 48(1): 16-31. [Crossref]
3. de Lucena Alves C, de Almeida S, Lima D, Neto P, Miranda AL, Manini T, et al. Muscle quality in older adults: a scoping review. J Am Med Dir Assoc, 2023, 24(4): 462-467.e412. [Crossref]
4. Virto N, Río X, Méndez-Zorrilla A, & García-Zapirain B. Non invasive techniques for direct muscle quality assessment after exercise intervention in older adults: a systematic review. BMC Geriatr, 2024, 24(1): 642. [Crossref]
5. Barbat-Artigas S, Rolland Y, Zamboni M, & AubertinLeheudre M. How to assess functional status: a new muscle quality index. J Nutr Health Aging, 2012, 16(1): 67-77. [Crossref]
6. Coronado-Zarco R, & de León A. Muscle quality an evolving concept. J Frailty Sarcopenia Falls, 2023, 8(4): 254-260. [Crossref]
7. Kuschel L, Sonnenburg D, & Engel T. Factors of muscle quality and determinants of muscle strength: a systematic literature review. Healthcare (Basel), 2022, 10(10): 1937-1948. [Crossref]
8. Distefano G, Standley R, Zhang X, Carnero E, Yi F, Cornnell H, et al. Physical activity unveils the relationship between mitochondrial energetics, muscle quality, and physical function in older adults. J Cachexia Sarcopenia Muscle, 2018, 9(2): 279-294. [Crossref]
9. Correa-de-Araujo R, Harris-Love M, Miljkovic I, Fragala M, Anthony B, & Manini T. The need for standardized assessment of muscle quality in skeletal muscle function deficit and other aging-related muscle dysfunctions: a symposium report. Front Physiol, 2017, 8: 87-95. [Crossref]
10. Chodzko-Zajko W, Proctor D, Fiatarone Singh M, Minson C, Nigg C, Salem GJ, et al. American College of Sports Medicine position stand. Exercise and physical activity for older adults. Med Sci Sports Exerc, 2009, 41(7): 1510-1530. [Crossref]
11. Baltzopoulos V, & Brodie D. Isokinetic dynamometry. Applications and limitations. Sports Med, 1989, 8(2): 101-116. [Crossref]
12. Niklasson E, Borga M, Dahlqvist Leinhard O, Widholm P, Andersson D, Wiik A, et al. Assessment of anterior thigh muscle size and fat infiltration using single-slice CT imaging versus automated MRI analysis in adults. Br J Radiol, 2022, 95(1133): 20211094. [Crossref]
13. Faron A, Sprinkart A, Kuetting D, Feisst A, Isaak A, Endler C, et al. Body composition analysis using CT and MRI: intra-individual intermodal comparison of muscle mass and myosteatosis. Sci Rep, 2020, 10(1): 11765. [Crossref]
14. Abdalla P, Bohn L, da Silva L, Dos Santos A, Tasinafo Junior M, Venturini A, et al. Identification of muscle weakness in older adults from normalized upper and lower limbs strength: a cross-sectional study. BMC Sports Sci Med Rehabil, 2021, 13(1): 161-173. [Crossref]
15. Abe T, Thiebaud R, & Loenneke J. Forearm muscle quality as a better indicator of physical performance than handgrip strength in older male ground golf players aged 70 to 89. J Musculoskelet Neuronal Interact, 2016, 16(4): 296-301.
16. Baek J, Jang I, Jung H, Park S, Lee J, Choi E, et al. Serum irisin level is independent of sarcopenia and related muscle parameters in older adults. Exp Gerontol, 2022, 162: 111744. [Crossref]
17. Banitalebi E, Faramarzi M, Ghahfarokhi M, SavariNikoo F, Soltani N, & Bahramzadeh A. Osteosarcopenic obesity markers following elastic band resistance training: A randomized controlled trial. Exp Gerontol, 2020, 135: 110884. [Crossref]
18. Barbat-Artigas S, Rolland Y, Vellas B, & Aubertin-Leheudre M. Muscle quantity is not synonymous with muscle quality. J Am Med Dir Assoc, 2013, 14(11): 852.e851-857. [Crossref]
19. Brady A, Straight C, Schmidt M, & Evans E. Impact of body mass index on the relationship between muscle quality and physical function in older women. J Nutr Health Aging, 2014, 18(4): 378-382. [Crossref]
20. Briggs R, Houck J, Drummond M, Fritz J, Lastayo P, & Marcus R. Muscle quality improves with extended highintensity resistance training after hip fracture. Journal of Frailty & Aging, 2018, 7(1): 51-56. [Crossref]
21. Brightwell C, Markofski M, Moro T, Fry C, Porter C, Volpi E, et al. Moderate-intensity aerobic exercise improves skeletal muscle quality in older adults. Transl Sports Med, 2019, 2(3): 109-119. [Crossref]
22. Brown J, Harhay M, & Harhay M. The muscle quality index and mortality among males and females. Ann Epidemiol, 2016, 26(9): 648-653. [Crossref]
23. Canon M, & Crimmins E. Sex differences in the association between muscle quality, inflammatory markers, and cognitive decline. J Nutr Health Aging, 2011, 15(8): 695-698. [Crossref]
24. Chang C, Lin C, Hsieh H, Lo W, Lai Y, Peng L, et al. Risk of sarcopenia among older persons with type 2 diabetes mellitus with different status of albuminuria: a doseresponsive association. Arch Gerontol Geriatr, 2021, 95: 104338. [Crossref]
25. Correa C, Baroni B, Radaelli R, Lanferdini F, Cunha Gdos S, Reischak-Oliveira Á, et al. Effects of strength training and detraining on knee extensor strength, muscle volume and muscle quality in elderly women. Age (Dordr), 2013, 35(5): 1899-1904. [Crossref]
26. da Costa Pereira J, Diniz A, de Lemos M, Pinho Ramiro C, & Cabral P. Frailty but not low muscle quality nor sarcopenia is independently associated with mortality among previously hospitalized older adults: a prospective study. Geriatr Gerontol Int, 2023, 23(10): 736-743. [Crossref]
27. Cramer J, Cruz-Jentoft A, Landi F, Hickson M, Zamboni M, Pereira S, et al. Impacts of high-protein oral nutritional supplements among malnourished men and women with sarcopenia: a multicenter, randomized, double-blinded, controlled trial. J Am Med Dir Assoc, 2016, 17(11): 1044-1055. [Crossref]
28. Cunha P, Nunes J, Tomeleri C, Nascimento M, Schoenfeld B, Antunes M, et al. Resistance training performed with single and multiple sets induces similar improvements in muscular strength, muscle mass, muscle quality, and IGF-1 in older women: a randomized controlled trial. J Strength Cond Res, 2020, 34(4): 1008-1016. [Crossref]
29. Delmonico M, Zmuda J, Taylor B, Cauley J, Harris T, Manini T, et al. Association of the ACTN3 genotype and physical functioning with age in older adults. J Gerontol A Biol Sci Med Sci, 2008, 63(11): 1227-1234. [Crossref]
30. Dos Santos V, Antunes M, Dos Santos L, Nascimento M, Pina F, Carneiro N, et al. Effects of different resistance training frequencies on body composition, muscular strength, muscle quality, and metabolic biomarkers in sarcopenic older women. J Strength Cond Res, 2024, 38(9): e521-e528. [Crossref]
31. Emerson N, Fukuda D, Stout J, Robinson E, McCormack W, Scanlon T, et al. Physical working capacity at fatigue threshold (PWCFT) is associated with sarcopeniarelated body composition and measures of functionality in older adults. Archives of Gerontology and Geriatrics, 2014, 59(2): 300-304. [Crossref]
32. Dalle S, Van Roie E, Hiroux C, Vanmunster M, Coudyzer W, Suhr F, et al. Omega-3 supplementation improves isometric strength but not muscle anabolic and catabolic signaling in response to resistance exercise in healthy older adults. J Gerontol A Biol Sci Med Sci, 2021, 76(3): 406-414. [Crossref]
33. Fragala M, Jajtner A, Beyer K, Townsend J, Emerson N, Scanlon T, et al. Biomarkers of muscle quality: N-terminal propeptide of type III procollagen and C-terminal agrin fragment responses to resistance exercise training in older adults. J Cachexia Sarcopenia Muscle, 2014, 5(2): 139-148. [Crossref]
34. Fragala M, Kenny A, & Kuchel G. Muscle quality in aging: a multi-dimensional approach to muscle functioning with applications for treatment. Sports Med, 2015, 45(5): 641-658. [Crossref]
35. Gadelha A, Neri S, Nóbrega O, Pereira J, Bottaro M, Fonsêca A, et al. Muscle quality is associated with dynamic balance, fear of falling, and falls in older women. Exp Gerontol, 2018, 104: 1-6. [Crossref]
36. Gadelha A, Neri S, Bottaro M, & Lima R. The relationship between muscle quality and incidence of falls in older community-dwelling women: an 18-month follow-up study. Exp Gerontol, 2018, 110: 241-246. [Crossref]
37. Ghasemikaram M, Engelke K, Kohl M, von Stengel S, & Kemmler W. Detraining effects on muscle quality in older men with osteosarcopenia. Follow-up of the randomized controlled franconian osteopenia and sarcopenia trial (FrOST). Nutrients, 2021, 13(5): 1528-1539. [Crossref]
38. Goodpaster B, Park S, Harris T, Kritchevsky S, Nevitt M, Schwartz A, et al. The loss of skeletal muscle strength, mass, and quality in older adults: the health, aging and body composition study. J Gerontol A Biol Sci Med Sci, 2006, 61(10): 1059-1064. [Crossref]
39. Hairi N, Cumming R, Naganathan V, Handelsman D, Le Couteur D, Creasey H, et al. Loss of muscle strength, mass (sarcopenia), and quality (specific force) and its relationship with functional limitation and physical disability: the Concord Health and Ageing in Men Project. J Am Geriatr Soc, 2010, 58(11): 2055-2062. [Crossref]
40. Herda A, & Nabavizadeh O. Short-term resistance training in older adults improves muscle quality: a randomized control trial. Exp Gerontol, 2021, 145: 111195. [Crossref]
41. Jerez-Mayorga D, Delgado-Floody P, Intelangelo L, Campos-Jara C, Arias-Poblete L, García-Verazaluce J, et al. Behavior of the muscle quality index and isometric strength in elderly women. Physiology & Behavior, 2020, 227: 113145. [Crossref]
42. Katsiaras A, Newman A, Kriska A, Brach J, Krishnaswami S, Feingold E, et al. Skeletal muscle fatigue, strength, and quality in the elderly: the Health ABC Study. J Appl Physiol (1985), 2005, 99(1): 210-216. [Crossref]
43. Kennis E, Verschueren S, Bogaerts A, Coudyzer W, Boonen S, & Delecluse C. Effects of fitness and vibration training on muscle quality: a 1-year postintervention follow-up in older men. Arch Phys Med Rehabil, 2013, 94(5): 910-918. [Crossref]
44. Nonaka K, Murata S, Shiraiwa K, Abiko T, Goda A, Yasufuku Y, et al. Correlations between physical performance and muscle mass, strength, and quality in obese and normal-weight community-dwelling older Japanese women. Asian Journal of Gerontology and Geriatrics, 2020, 15: 17-22. [Crossref]
45. Koster A, Ding J, Stenholm S, Caserotti P, Houston D, Nicklas B, et al. Does the amount of fat mass predict agerelated loss of lean mass, muscle strength, and muscle quality in older adults? J Gerontol A Biol Sci Med Sci, 2011, 66(8): 888-895. [Crossref]
46. Liao C, Tsauo J, Huang S, Ku J, Hsiao D, & Liou T. Effects of elastic band exercise on lean mass and physical capacity in older women with sarcopenic obesity: A randomized controlled trial. Sci Rep, 2018, 8(1): 2317-2328. [Crossref]
47. Lopez P, Crosby B, Robetti B, Turella D, Weber T, de Oliveira M, et al. Effects of an 8-week resistance training intervention on plantar flexor muscle quality and functional capacity in older women: a randomised controlled trial. Exp Gerontol, 2020, 138: 111003. [Crossref]
48. Maltais M, Boisvert-Vigneault K, Rolland Y, Vellas B, & de Souto Barreto P. Longitudinal associations of physical activity levels with morphological and functional changes related with aging: The MAPT study. Exp Gerontol, 2019, 128: 110758. [Crossref]
49. McCormack W, Stout J, Emerson N, Scanlon T, Warren A, Wells A, et al. Oral nutritional supplement fortified with beta-alanine improves physical working capacity in older adults: a randomized, placebo-controlled study. Exp Gerontol, 2013, 48(9): 933-939. [Crossref]
50. Michel E, Zory R, Guerin O, Prate F, Sacco G, & Chorin F. Assessing muscle quality as a key predictor to differentiate fallers from non-fallers in older adults. Eur Geriatr Med, 2024, 15(5): 1301-1311. [Crossref]
51. Misic M, Rosengren K, Woods J, & Evans E. Muscle quality, aerobic fitness and fat mass predict lower-extremity physical function in community-dwelling older adults. Gerontology, 2007, 53(5): 260-266. [Crossref]
52. Moore A, Caturegli G, Metter E, Makrogiannis S, Resnick S, Harris T, et al. Difference in muscle quality over the adult life span and biological correlates in the Baltimore Longitudinal Study of Aging. J Am Geriatr Soc, 2014, 62(2): 230-236. [Crossref]
53. Müller D, Izquierdo M, Boeno F, Aagaard P, Teodoro J, Grazioli R, et al. Adaptations in mechanical muscle function, muscle morphology, and aerobic power to highintensity endurance training combined with either traditional or power strength training in older adults: a randomized clinical trial. Eur J Appl Physiol, 2020, 120(5): 1165-1177. [Crossref]
54. Neri S, Pereira J, de David A, & Lima R. The influence of body fat distribution on postural balance and muscle quality in women aged 60 years and over. J Appl Biomech, 2021, 37(3): 182-187. [Crossref]
55. Neri S, Harvey L, Tiedemann A, Gadelha A, & Lima R. Obesity and falls in older women: mediating effects of muscle quality, foot loads and postural control. Gait Posture, 2020, 77: 138-143. [Crossref]
56. Neto I, Diniz J, Alves V, Ventura Oliveira A, Barbosa M, da Silva Prado C, et al. Field-based estimates of muscle quality index determine timed-up-and-go test performance in obese older women. Clin Interv Aging, 2023, 18: 293-303. [Crossref]
57. de Sousa Neto I, da Cunha Nascimento D, Prestes J, da Fonseca E, Celes R, Rolnick N, et al. Initial muscle quality affects individual responsiveness of interleukin-6 and creatine kinase following acute eccentric exercise in sedentary obese older women. Biology (Basel), 2022, 11(4): 537-548. [Crossref]
58. Nogueira Paranhos Amorim D, Nascimento D, Stone W, Alves V, Moraes C, & Coelho Vilaça E. Muscle quality is associated with history of falls in octogenarians. J Nutr Health Aging, 2021, 25(1): 120-125. [Crossref]
59. Nunes J, Ribeiro A, Silva A, Schoenfeld B, Santos L, Cunha P, et al. Improvements in phase angle are related with muscle quality index after resistance training in older women. Journal of Aging and Physical Activity, 2018: 1-19. [Crossref]
60. Oh S, Kim H, Woo S, Cho B, Song M, Park Y, et al. Effects of an integrated health education and elastic band resistance training program on physical function and muscle strength in community-dwelling elderly women: Healthy Aging and Happy Aging II study. Geriatr Gerontol Int, 2017, 17(5): 825-833. [Crossref]
61. Osuka Y, Kojima N, Nishihara K, Sasai H, Wakaba K, Tanaka K, et al. β-hydroxy-β-methylbutyrate supplementation may not enhance additional effects of exercise on muscle quality in older women. Med Sci Sports Exerc, 2022, 54(4): 543-550. [Crossref]
62. Park S, Goodpaster B, Strotmeyer E, de Rekeneire N, Harris T, Schwartz A, et al. Decreased muscle strength and quality in older adults with type 2 diabetes: the health, aging, and body composition study. Diabetes, 2006, 55(6): 1813-1818. [Crossref]
63. Pancera S, Lopomo N, Porta R, Sanniti A, Buraschi R, & Bianchi L. Effects of combined endurance and resistance eccentric training on muscle function and functional performance in patients with chronic obstructive pulmonary disease: randomized controlled trial. Arch Phys Med Rehabil, 2024, 105(3): 470-479. [Crossref]
64. Pinto R, Correa C, Radaelli R, Cadore E, Brown L, & Bottaro M. Short-term strength training improves muscle quality and functional capacity of elderly women. Age (Dordr), 2014, 36(1): 365-372. [Crossref]
65. Radaelli R, Brusco C, Lopez P, Rech A, Machado C, Grazioli R, et al. Muscle quality and functionality in older women improve similarly with muscle power training using one or three sets. Exp Gerontol, 2019, 128: 110745. [Crossref]
66. Radaelli R, Wilhelm E, Botton C, Rech A, Bottaro M, Brown L, et al. Effects of single vs. multiple-set shortterm strength training in elderly women. Age (Dordr), 2014, 36(6): 9720-9726. [Crossref]
67. Rava A, Pihlak A, Ereline J, Gapeyeva H, Kums T, Purge P, et al. Body composition, neuromuscular performance, and mobility: comparison between regularly exercising and inactive older women. J Aging Phys Act, 2017, 25(1): 58-64. [Crossref]
68. Reinders I, Murphy R, Koster A, Brouwer I, Visser M, Garcia M, et al. Muscle quality and muscle fat infiltration in relation to incident mobility disability and gait speed decline: the age, gene/environment susceptibility-reykjavik study. J Gerontol A Biol Sci Med Sci, 2015, 70(8): 1030-1036. [Crossref]
69. Ribeiro A, Picoloto A, Nunes J, Bezerra E, Schoenfeld B, & Cyrino E. Effects of different resistance training loads on the muscle quality index in older women. J Strength Cond Res, 2022, 36(5): 1445-1449. [Crossref]
70. Ribeiro A, Schoenfeld B, Souza M, Tomeleri C, Venturini D, Barbosa D, et al. Traditional and pyramidal resistance training systems improve muscle quality and metabolic biomarkers in older women: a randomized crossover study. Exp Gerontol, 2016, 79: 8-15. [Crossref]
71. Rondanelli M, Guido D, Opizzi A, Faliva M, Perna S, & Grassi M. A path model of sarcopenia on bone mass loss in elderly subjects. J Nutr Health Aging, 2014, 18(1): 15-21. [Crossref]
72. Salas-Groves E, Alcorn M, Childress A, & Galyean S. The effect of web-based culinary medicine to enhance protein intake on muscle quality in older adults: randomized controlled trial. JMIR Form Res, 2024, 8: e49322. [Crossref]
73. Schroeder E, He J, Yarasheski K, Binder E, CastanedaSceppa C, Bhasin S, et al. Value of measuring muscle performance to assess changes in lean mass with testosterone and growth hormone supplementation. Eur J Appl Physiol, 2012, 112(3): 1123-1131. [Crossref]
74. Seo M, Jung S, Kim S, Jung H, Kim D, & Song J. Comparisons of muscle quality and muscle growth factor between sarcopenic and non-sarcopenic older women. Int J Environ Res Public Health, 2020, 17(18): 6581-6593. [Crossref]
75. Shin S, Valentine R, Evans E, & Sosnoff J. Lower extremity muscle quality and gait variability in older adults. Age Ageing, 2012, 41(5): 595-599. [Crossref]
76. Silva P, Nascimento D, IV D, Funghetto S, Tibana R, Navalta J, et al. Effects of resistance training on muscle quality index, muscle strength, functional capacity, and serum immunoglobulin levels between obese and nonobese older women. Int J Exerc Sci, 2021, 14(7): 707-726. [Crossref]
77. Spira D, Walston J, Buchmann N, Nikolov J, Demuth I, Steinhagen-Thiessen E, et al. Angiotensin-converting enzyme inhibitors and parameters of sarcopenia: relation to muscle mass, strength and function: data from the Berlin Aging Study-II (BASE-II). Drugs Aging, 2016, 33(11): 829-837. [Crossref]
78. Stotz A, Mason J, Groll A, & Zech A. Which trunk muscle parameter is the best predictor for physical function in older adults? Heliyon, 2023, 9(10): e20123. [Crossref]
79. Straight C, Brady A, & Evans E. Muscle quality and relative adiposity are the strongest predictors of lowerextremity physical function in older women. Maturitas, 2015, 80(1): 95-99. [Crossref]
80. Sui S, Williams L, Holloway-Kew K, Hyde N, Leach S, & Pasco J. Associations between muscle quality and cognitive function in older men: cross-sectional data from the Geelong Osteoporosis Study. Journal of Clinical Densitometry, 2022, 25(2): 133-140. [Crossref]
81. Tracy B, Ivey F, Hurlbut D, Martel G, Lemmer J, Siegel E, et al. Muscle quality. II. Effects of strength training in 65- to 75-yr-old men and women. J Appl Physiol (1985), 1999, 86(1): 195-201. [Crossref]
82. Vilaça K, Carneiro J, Ferriolli E, Lima N, de Paula F, & Moriguti J. Body composition, physical performance and muscle quality of active elderly women. Arch Gerontol Geriatr, 2014, 59(1): 44-48. [Crossref]
83. Vilaça K, Alves N, Carneiro J, Ferriolli E, Lima N, & Moriguti J. Body composition, muscle strength and quality of active elderly women according to the distance covered in the 6-minute walk test. Braz J Phys Ther, 2013, 17(3): 289-296. [Crossref]
84. Virto N, Río X, Muñoz I, Méndez-Zorrilla A, & García-Zapirain B. Gait speed in older adults: exploring the impact of functional, physical and social factors. Retos, 2024, 61: 552-566. [Crossref]
85. Vojciechowski A, Silva C, Rodrigues E, Gallo L, Melo Filho J, & Gomes A. Does physical dance training with virtual games change muscle quality of community-dwelling older women? Games Health J, 2021, 10(6): 391-399. [Crossref]
86. Volpato S, Bianchi L, Lauretani F, Lauretani F, Bandinelli S, Guralnik J, et al. Role of muscle mass and muscle quality in the association between diabetes and gait speed. Diabetes Care, 2012, 35(8): 1672-1679. [Crossref]
87. Walsh G, Low D, & Arkesteijn M. The relationship between postural control and muscle quality in older adults. J Mot Behav, 2022, 54(3): 363-371. [Crossref]
88. Yamada M, Kimura Y, Ishiyama D, Nishio N, Abe Y, Kakehi T, et al. Differential characteristics of skeletal muscle in community-dwelling older adults. J Am Med Dir Assoc, 2017, 18(9): 807.e809-807.e816. [Crossref]
89. Yamaguchi Y, Greiner C, Lee S, Ryuno H, Yen H, Lin C, et al. Lifestyle factors associated with muscle quality in community-dwelling older people with type 2 diabetes in Japan and Taiwan: a cross-sectional study. Psychogeriatrics, 2022, 22(5): 736-742. [Crossref]
90. Yoon J, Ha Y, Kim K, Moon J, Choi S, Lim S, et al. Hyperglycemia is associated with impaired muscle quality in older men with diabetes: the Korean Longitudinal Study on Health and Aging. Diabetes Metab J, 2016, 40(2): 140-146. [Crossref]
91. Yu X, Wu D, Li F, Qiao W, & Chen X. Correlation between intrinsic capacity and muscle strength and quality in older patients with cardiovascular disease: a crosssectional study. Clin Interv Aging, 2024, 19: 1703-1711. [Crossref]
92. Stark T, Walker B, Phillips J, Fejer R, & Beck R. Hand-held dynamometry correlation with the gold standard isokinetic dynamometry: a systematic review. Pm r, 2011, 3(5): 472-479. [Crossref]
93. Li R, Jasiewicz J, Middleton J, Condie P, Barriskill A, Hebnes H, et al. The development, validity, and reliability of a manual muscle testing device with integrated limb position sensors. Arch Phys Med Rehabil, 2006, 87(3): 411-417. [Crossref]
94. Grootswagers P, Vaes A, Hangelbroek R, Tieland M, van Loon L, & de Groot L. Relative validity and reliability of isometric lower extremity strength assessment in older adults by using a handheld dynamometer. Sports Health, 2022, 14(6): 899-905. [Crossref]
95. Grgic J, Lazinica B, Schoenfeld B, & Pedisic Z. Testretest reliability of the one-repetition maximum (1RM) strength assessment: a systematic review. Sports Med Open, 2020, 6(1): 31-43. [Crossref]
96. Bassey E, & Short A. A new method for measuring power output in a single leg extension: feasibility, reliability and validity. Eur J Appl Physiol Occup Physiol, 1990, 60(5): 385-390. [Crossref]
97. Albano D, Messina C, Vitale J, & Sconfienza L. Imaging of sarcopenia: old evidence and new insights. Eur Radiol, 2020, 30(4): 2199-2208. [Crossref]
98. Sconfienza L, Albano D, Allen G, Bazzocchi A, Bignotti B, Chianca V, et al. Clinical indications for musculoskeletal ultrasound updated in 2017 by European Society of Musculoskeletal Radiology (ESSR) consensus. Eur Radiol, 2018, 28(12): 5338-5351. [Crossref]
99. Perkisas S, Baudry S, Bauer J, Beckwée D, De Cock A, Hobbelen H, et al. Application of ultrasound for muscle assessment in sarcopenia: towards standardized measurements. Eur Geriatr Med, 2018, 9(6): 739-757. [Crossref]
100. Beaudart C, Bruyère O, Geerinck A, Hajaoui M, Scafoglieri A, Perkisas S, et al. Equation models developed with bioelectric impedance analysis tools to assess muscle mass: a systematic review. Clin Nutr ESPEN, 2020, 35: 47-62. [Crossref]
SUPPLEMENTARY
Table S1.
Representative MQ metrics.
| Study | Participants | MQ category | MQ formula | Assessment methods |
|---|---|---|---|---|
| Barbat et al. [18] (Canada) | 1219 women (80 ± 4 yrs) | Strength-to-mass ratio | Muscle Quality Index (MQI): strength per unit of muscle mass Upper limb: handgrip strength by upper limbs muscle mass (kPa/kg). Lower limb: KES by lower limbs muscle mass (kPa/kg) |
Test: HGS and KES test Instrument: For muscle strength: isometric dynamometer (HGS) and maximum voluntary contraction for (KES); for muscle mass and body composition, DXA |
| Brown et al. [22] (USA) | 4510 older adults in a longitudinal cohort study (≥60 yrs) | Field-test surrogate measure | MQI: Based on sit-to-stand time, body mass, and leg length. [(L - 0.5) × body mass × g × 5]/T sit to stand; where 0.5 (m), Leg (m), g (m/s2) |
Test: 5-repetition sit-to-stand Instrument: For functional strength: time analysis of 5 sit to stand test; anthropometry for leg length and digital scale for body mass |
| Delmonico et al. [29] (USA) | 1367 well-functioning older adults (726 men, 70–79 yrs) | Strength-to-morphology ratio | MQ, knee extensor torque per unit of thigh cross sectional area | Test: Knee extensor torque Instrument: For muscle strength, Kin-Com dynamometer for isokinetic strength; CT for CSA measurement |
| Fragala et al. [34] (USA) | 25 older adults (70.6 ± 6.1 yrs) | Field-test surrogate measure | Muscle Quality Index (MQI) based on sit-to-stand power, body mass, and leg length MQI= ((leg length × 0.4) × body mass × gravity × 10)/ time sit to stand |
Test: 5 Sit-to-stand (STS). Leg length Instrument: STS performance; for body mass and leg length DXA |
| Gadelha et al. [35] (Brazil) | 246 community-dwelling older women (68.1 ± 6.2 yrs) | Strength-to-morphology ratio | Peak torque per unit of thigh muscle thickness | Test: Knee extension Instrument: Isokinetic dynamometer for dominant leg strength; B-mode ultrasound for muscle thickness |
| Goodpaster et al. [38] (USA) | 1880 older adults (73.5 ± 2.8 yrs, 51.6% women) | Strength-to-mass ratio | Isokinetic torque per unit of leg lean mass (Nm/kg) | Test: Knee extensor isokinetic strength Instrument: For muscle strength; Kin-Com dynamometer (isokinetic strength); for muscle mass: DXA |
| Hairi et al. [39] (Australia) | 1705 older men (≥70 yrs) | Strength-to-mass ratio | MQ, ratio of strength per unit of mass in lower and upper extremities | Test: HGS and quadriceps muscle quadriceps strength. Instrument: For muscle strength: Jamar isometric dynamometer, spring gauge on both legs separately (lower limbs limb muscle); for muscle mass, DXA |
| Katsiaras et al. [42] (USA) | 1512 older adults (70–79 yrs) | Strength-to-morphology ratio | Muscle-specific torque, that is, peak torque per cross-sectional area, was calculated as an index of muscle quality. (strength/CSA) |
Test: Knee extensors and flexors Instrument: For muscle strength, Isokinetic dynamometer in knee extensors and flexors; for CSA CT |
| Maltais et al. [48] (France) | 1679 older adults (75.3 ± 4.4 yrs) | Strength-to-mass ratio | 1. MQ, handgrip strength per unit of body weight (kg/kg) 2. MQ, handgrip strength divided by arm fat free mass (kg/kg) |
Test: HGS Instrument: For muscle strength: isometric dynamometer (HGS); for muscle mass DXA (body composition) |
| Moore et al. [52] (USA) | 786 adults (66.3 yrs) | Strength-to-morphology ratio | MQ, knee-extension strength per muscle cross-sectional area | Test: Knee extensors Instrument: For muscle strength, Isokinetic dynamometer in knee extensors and flexors; for CSA CT |
| Müller et al. [53] (Germany) | 35 older men (65.8 ± 3.9 yrs) | Strength-to-morphology ratio. | MQ, specific tension formula, ratio between the knee extension 1RM/2 and the sum of the squared quadriceps musclequadriceps femoris muscles (N/cm2). | Test: Knee extension Instrument: For muscle strength, 1RM test in knee extension; for quadriceps musclequadriceps femoris volume B-mode ultrasound derived muscle thickness |
| Park et al. [62] (USA) | 2618 older adults (70-79 yrs) | Strength-to-mass ratio | MQ; muscle strength per regional muscle mass (Nm/kg). Leg specific force/leg muscle mass or Arm specific force/ arm muscle mass |
Test: HGS and knee extension Instrument: For muscle strength: isometric dynamometer (HGS) and isokinetic dynamometer for KES; for leg or arm muscle mass DXA |
| Tracy et al. [81] (USA) | 23 older adults (65-75 yrs) | Strength-to-morphology ratio | MQ, strength per unit of muscle mass (muscle volume) (kg/cm3) (cm3). Isometric and 1-RM strength values (N and kg, respectively)/ muscle volume | Test: Knee extension Instrument: For muscle strength: 1RM strength test, isometric force production, and isokinetic peak torque were assessed in the knee extensors; for muscle mass and volume: MRI |
| Vojciechowski et al. [85] (Brazil) | 46 older women (69 ± 4 yrs) | Strength-to-morphology ratio | MQ, quadriceps musclequadriceps peak torque to cross-sectional area (CSA) | Test: Knee extension, knee flexion, ankle dorsiflexion and ankle plantarflexion Instrument: For muscle strength, isokinetic dynamometer for knee extension, knee flexion, ankle dorsiflexion and ankle plantarflexion test; for cross sectional area MRI |
| Yamada et al. [88] (Japan) | 347 healthy older adults (81 ± 7 yrs, 247 women) | Strength-to-morphology ratio | MQ, knee extension torque divided by the quadriceps musclequadriceps femoris muscle thickness (Nm/cm) | Test: Knee extension Instrument: For muscle strength, isokinetic dynamometer for knee extension; for quadriceps musclequadriceps femoris volume B-mode ultrasound derived muscle thickness |