Open Access | Research Article
This work is licensed under a
Creative Commons Attribution-ShareAlike 4.0
International License.
Study of surgical site infection: An obstetrical surgical morbidity at a tertiary level hospital
* Correspondence to: Deepika Panwar
Mailing address:
Department of Obstetrics and Gynecology, Umaid Hospital, Dr. S.N. Medical
College and Hospital, Jodhpur, India.
E-mail:
panwardeepika19@gmail.com
Received: 3 April 2021 / Accepted: 12 July 2021
DOI:
10.31491/CSRC.2021.09.078
Abstract
Background: Surgical site infection (SSI) is amongst the most
common sufferings following cesarean section. It contributes to
increased morbidity and negative impact on the mental, social and
economic condition of patients. This study aimed to determine the
incidence, risk factors, and therefore the bacteriological profile
following cesarean section at Umaid Hospital Jodhpur.
Methods: This was a hospital-based prospective observational
study of 1600 patients who had cesarean section over 3 months
duration. Among them, 50 patients developed post-cesarean SSI. Wound
swabs were collected from these patients. Culture and antibiotic
sensitivity were done for aerobic pyogenic organisms.
Results: Out of the 1600 participants who had a cesarean
section, 50 patients had SSI, giving an incidence of 3.12 of 100
cesarean sections. The common isolates were coagulase-negative
Staphylococcus Aureus (57%), Staphylococcus Aureus (14%), E.coli
(17%), Acinetobacter (7%), and Klebsiella (3%). The risk factors were
emergency cesarean section, obesity, rupture of membranes, lack of
intraoperative antibiotic coverage, previous cesarean section, etc.
The common isolates were resistant to Ofloxacin and sensitive to
Vancomycin, Linezolid, and Amikacin.
Conclusion: The post-cesarean wound infection rate in our
center was 3.12 of 100 cesarean sections. Linezolid, Cefazoline
antibiotics were sensitive for the common isolates from SSI and may be
used prophylactically till the final report of culture and sensitivity
is obtained. This may reduce the complications associated with SSI.
Keywords
Cesarean section, surgical site infection, wound infections, antibiotic sensitivity
Introduction
Surgical site infection (SSI) is defined as an infection occurring
within 30 days after surgery and affecting superficial/deep tissues at
the operation site [1]. SSI is one of the most common causes
of nosocomial infections, with a reported incidence rate of 2-20%
[2]. Postoperative SSI following cesarean section is related
to increased morbidity, mortality, prolonged hospital stays, and
socio-economic loss to the patients [3].
Among risk factors,
patient-related factors are old age, nutritional status, pre-existing
infection, co-morbid illness, and procedure-related factors like poor
surgical technique, prolonged duration of surgery, pre-operative part
preparation, and improper aseptic precautions. These factors can
influence SSI significantly [2].
In addition to these risk
factors, the virulence and the invasive power of the organism involved,
the physiological state of the wound tissue, and the immunological
integrity of the host are also important. SSI may delay the recovery of
patients, prolong hospital stay or outpatient treatment, necessitate
readmission, and lead to significant morbidity and mortality
[4]. The rate of SSI after cesarean section ranges from 3% to
15% in different settings [5-7].
The underlying
predisposing factors for SSI following cesarean include intrinsic
factors like age, obesity, medical conditions like diabetes mellitus and
hypertension, and immune-compromised statuses like HIV infection and
anemia [8-11]. Extrinsic factors identified in previous
studies include preoperative part preparation, type of procedure carried
out (emergency), type of skin incision given (horizontal/vertical),
prophylactic antibiotic coverage, chorioamnionitis, number of vaginal
examinations carried out before surgery, duration of operation, and
environment of the operating room [12-13]. The knowledge of
risk factors may help to reduce the incidence and severity of SSI.
The
CDC describes three levels of SSI; Superficial incisional SSI occurs
within 30 days after the operation, and the infection involves only skin
or subcutaneous tissue of the incision and at least one of the
following: 1. Purulent drainage, with or without laboratory
confirmation, from the superficial incision. 2. Organisms isolated from
an aseptically obtained culture of fluid or tissue from the superficial
incision. 3. At least one of the following signs or symptoms of
infection: pain or tenderness, localized swelling, redness or heat, and
the superficial incision is deliberately opened by the surgeon unless
the incision is culture-negative. 4. Diagnosis of superficial incisional
SSI by the surgeon or attending physician. Deep incisional SSI
Operationrelated infection involving deep soft tissues which occur
within 30 days after the operation and at least one of the following: 1.
Purulent drainage from the deep incision but not from the organ/space
component of the surgical site. 2. A deep incision spontaneously
dehisces or is deliberately opened by a surgeon when the patient has at
least one of the following signs or symptoms: fever (> 38 °C), localized
pain or tenderness unless the site is culture-negative. 3. An abscess or
other evidence of infection involving the deep incision is found on
direct examination, during reoperation, or by histopathologic or
radiologic examination. 4. Diagnosis of a deep incisional SSI by a
surgeon or attending physician [14].
Materials and methods
This hospital-based prospective observational study was carried out in the Department of Obstetrics and Gynecology at one of the largest tertiary care levels, Mother and Child Hospital, of western India. The study population comprised 1600 women who delivered by cesarean section over 3 months. A structured questionnaire was used to collect information from study subjects. Information was collected about demographic data, existing chronic diseases (such as diabetes mellitus and hypertension), and wound-related characteristics. All females were examined post-operatively for SSI features like purulent drainage from incision site till discharge from the hospital. Females who were readmitted with clinical features of SSI within 30 days of cesarean section were also included for calculation of SSI incidence. Two swabs were collected from the infection site using standard aseptic precautions and sent to the microbiology laboratory for further testing. In the laboratory, one swab was used for direct microscopy and Gram’s staining, from another swab aerobic pyogenic culture and sensitivity testing was done. Identification of organism and culture sensitivity reporting was done according to CLSI guidelines [15]. For detection of sepsis markers i.e., CRP and PCT, 3 mL of blood samples were collected in the plain vial, and the testing was done using Expedia Latex Agglutination and Dx Instant Check Kits, respectively. Due clearance was obtained from Institute, Ethics Committee (IEC No. SNMC/IEC/2021/plan/387). Written informed consent was taken from all subjects before inclusion into the study. Qualitative variables will be expressed as numbers and percentages and analyzed using the chi-square test.
Results
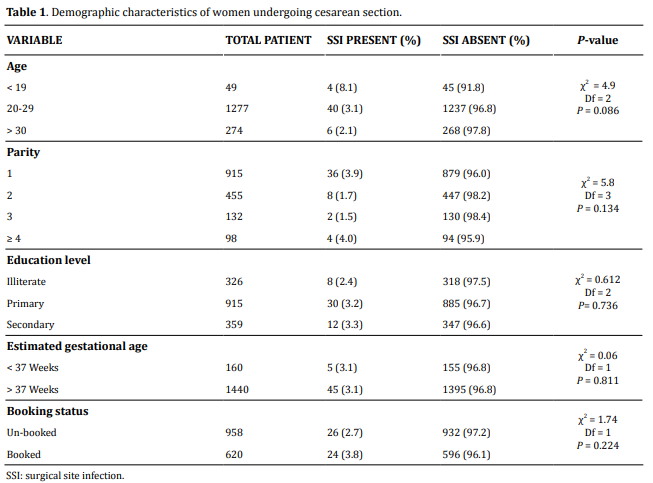
The study was performed for 3 months. Out of the 1600 women delivered by cesarean section, 50 (3.12%) women developed SSI (Figure 1). The proportion of SSI was the highest among teenagers (8.1%), among those with ≥ 4 children (4%), and those who had secondary education (3.3%) (Table 1).

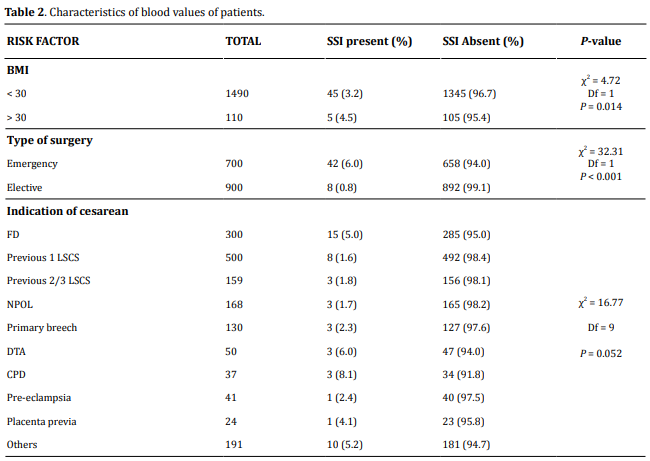
SSI was significantly higher among emergency cesarean section (6%) than elective cesarean section (0.8%) (P < 0.0001). Also, obese women (BMI> 30) were having more SSI (4.5%) than BMI < 30 (3.2%) (P=0.014). The most common indication of cesarean section developing SSI was fetal distress (5%) followed by previous LSCS. Rupture of membranes before cesarean section (2.8%) was associated with a higher risk of developing SSI than intact membranes (2.2%). Other factors associated with increased risk of post-cesarean wound infection were intra-operative blood loss greater than 500 mL (P < 0.001) and lack of intra-operative antibiotic prophylaxis (P < 0.001). Hospital stay was found to be higher in women developing SSI (100%) (Table 2).
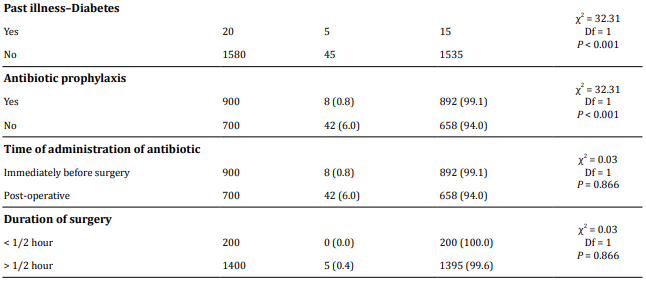

A total of 50 wound swabs were collected from patients developing post-cesarean SSI. Among these 28 (56%) had bacterial growth while 22 (44%) showed no growth (Figure 2).
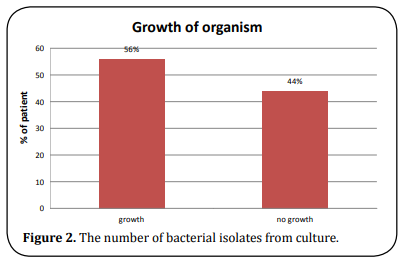
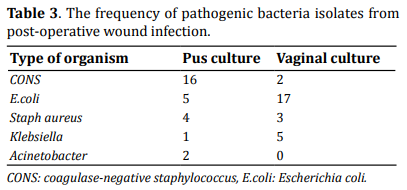
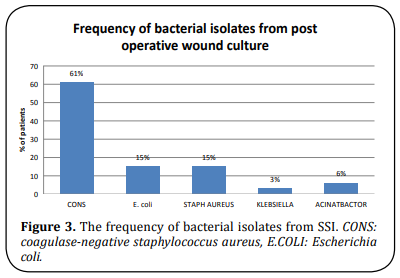
Table 3 shows the frequency of pathogenic bacteria isolates from post-operative wound infection. The most common organism isolated was CONS (61%) and the least common was Klebsiella (3%) (Figure 3). Images have been obtained by culturing the organism in the microbiology lab of Umaid Hospital (Figure 4).

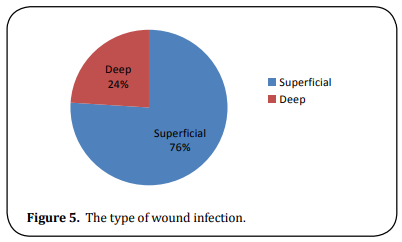
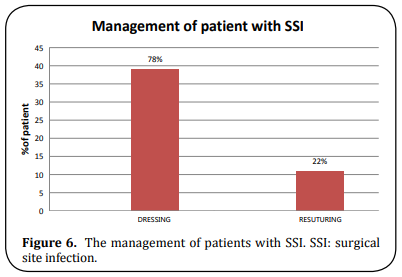
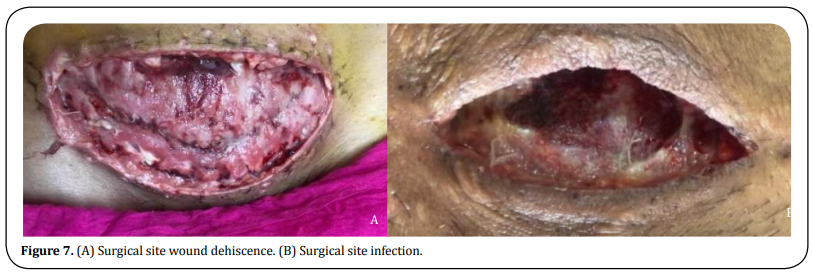
About 76% were superficial and 24% were deep wound infections (Figure 5). About 39 patients (78%) were managed by dressing alone while 11 (22%) required re-suturing (Figure 6). Images have been clicked in Umaid hospital (Figure 7).
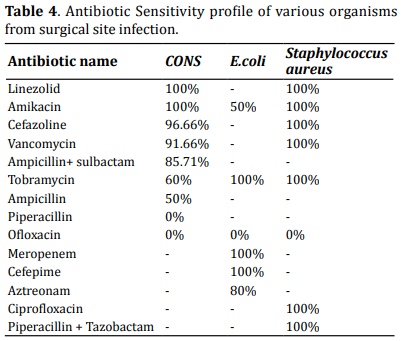
Among the organisms isolated from wound culture, most of the CONS (coagulase-negative staph aureus) were highly sensitive to Linezolid (100%) and Amikacin (100%), while highly resistant to Ofloxacin. E.coli was highly sensitive to Meropenem (100%), Cefepime (100%), and Tobramycin (100%) (Table 4).
Discussion
The study aimed to determine the incidence of postcesarean section wound
infection and the causative pathogens with their sensitivity profiles.
The incidence of post-cesarean wound infection in the present study was
3.12 per 100 cesarean sections. Similar past studies had reported an
incidence ranging from 7.8% to 8.5% [16]. The possible reason
for variation in these studies could be due to differences in the
population under study and the diversity of indications for cesarean
sections performed in different centers. The low incidence of SSI in the
present study could also be due to proper aseptic precautions being
followed at our institute.
In the present study, teenagers (< 19 years) were found to have a higher
proportion of SSI (8.1%). In a similar study, Cunningham et al. reported that many obstetrical complications
such as prolonged labor, PIH, and postpartum sepsis were more frequently observed among teenagers
[17].
In the present study, SSI was observed more among booked women compared
to un-booked. This could be due to extended hospital stay, nosocomial,
iatrogenic infection due to multiple per vaginal examinations, and
obstetric interventions in these patients as compared to the un-booked
patients, who were referred either delivered or operated relatively
early due to emergent indication.
In this study, SSI was more
common in educated women, which is contrary to findings of Njoku et al.
where SSI was seen more in uneducated women [18]. This may be
due to the reason that educated women were booked and were admitted for
an extended duration in the ward and had frequent obstetric examinations
by various strata of healthcare services.
The findings of this
study demonstrate a significant association between SSI and BMI. Obese
women (BMI > 30 Kg/m2) developed more SSI (4.5%) than those with BMI < 30 Kg/m2 (3.2%) (P=0.014). This finding
was similar to other studies [19]. It is because of relatively poor perfusion of adipose
tissue which may impair wound healing and decrease the local immune
response, enabling infection to occur. The incision for obese women may
also need to be longer and therefore involve more tissue becoming
exposed to contamination.
Generally, patients undergoing emergency
cesarean section are at higher risk of infections. In this study,
elective surgeries developing SSI were 6% while 0.8% were emergencies.
This is probably due to inadequate preparation time owing to
maternal/fetal distress, reduced attention to infection-preventing
procedures like prophylactic antibiotics, and increased urgency of the
procedure. Similar results were found in a study done by Njoku et al.
with SSI developing more in emergency cesarean sections as compared to
elective ones [17].
The common indication of cesarean
section developing SSI in our study was fetal distress (5%) followed by
previous LSCS. A study done by Wendmagegn et al. also showed fetal
distress to be the most common indication [20]. In our study,
76% of wound infections were superficial while 24% were deep wounds.
While a study done by Ghirmay et al. in 2015 showed superficial
incidence as 25% and deep 75% [21].
Prolonged duration
of surgery results in increased exposure of operation site to air,
prolonged trauma, prolonged anesthesia, and more blood loss. In our
study, 0.35% of patients operated on for > 1/2 hour developed SSI.
Shapiro et al. reported that with each hour of surgery the infection
rate almost doubles [22]. This finding was not significant
since cesarean section being routine obstetric surgery is completed in a
short duration (
< half hour).
In our study, blood loss of more than 500 mL was seen in 5% of cases
developing SSI. The risk of SSI rises by 30% for every 100 mL of blood
loss. A high volume of blood loss is usually associated with poor
control of bleeding, increased tissue damage, and more sutures
[23].
In this study, the type of suture material used in
surgery was not found to be significant as all the surgeries were done
using delayed absorbable suture material (chromic catgut and polyglactin
910).
In this study, out of 50 patients developing SSI, 28 patients
(56%) had microbial culture growth where Gram-positive cocci
(Staphylococcus aureus and CONS) was isolated in 72% cases, followed by
E.coli (17%), Acinetobacter sp. (7%), and Klebsiella sp. (3%) (Table 3).
Similar results were found in other studies done by Njoku et al. where
Staphylococcus aureus was the most common organism associated with SSI
[18].
In this study, the vaginal swab of patients
developing SSI was also sent, in which E.coli (62%) was the most common
bacteria following Klebsiella sp. (18%). The results were not found to
be significant as E.coli is a natural commensal of the vagina. Blood
samples for CRP (C-reactive protein) and PCT (procalcitonin) were also
collected but results were not significant as out of 50 patients
developing SSI, 45 (90%) were CRP positive and PCT was in the normal
range in all 50 patients. In this study, CONS isolates were sensitive to
Amikacin, Linezolid, Cefazoline, and Vancomycin (Table 4). Another study
done by Njoku et al. reported CONS to be sensitive to Amikacin and
Imipenem, and resistant to Cephalosporins, Amoxicillin/Clavulanate,
Gentamicin and Meropenem, and Fluoroquinolones [18]. Despite
the availability of antibiotics, SSI is still responsible for much
morbidity and socio-economic loss for both patients as well as health
care systems. Reduction in SSI while minimizing antibiotic resistance
remains a challenge for many health care institutions.
The
secondary resuturing rate was less at our center (22% underwent
resuturing while 78% of cases were managed on a conservative basis i.e.,
dressing alone) which shows that proper aseptic precautions were taken
while managing these patients and the secondary resuturing rate was
less.
SSI is known to cause prolonged hospital stay thus bringing
financial burden to the patient. In our study, all patients with SSI had
a hospital stay of > 7 days (100%) compared to others who were
discharged within 7 days of surgery.
The limitation of this study
was the lack of follow-up of patients who developed SSI after discharge
from the hospital within 30 days of surgery and went to other institutes
for management.
Conclusion
The incidence of SSI in this study was 3.12 per 100 cesarean sections.
The presence of wound sepsis was associated with a
longer duration of hospital stay which further led to economic loss to the patient.
Recommendations
1. Hand hygiene and infection prevention practice by health care providers have to be followed to reduce the risk of wound sepsis.
2. To evaluate and improve pre- and post-operative care, there is a need for continuous training and supervision of infection control practices.
3. Intraoperative antibiotics can be given to all patients scheduled for any obstetric intervention and cesarean section whether emergency/elective. Every institute should follow its antibiotic use protocols.
4. If a patient is diabetic, frequent and regular blood
sugar monitoring along with a low glycemic index diet and appropriate exercise.
5. A vigilant infection control committee should be
esstablished which should monitor SSI through surveillance studies with feedback data to healthcare workers, labor room, operation theatre, and post-operative staff, and surgeons are an important component of strategies to reduce the risk of SSI to a minimum acceptable level.
Declarations
Authors’ contributions
This study was contributed by all authors.
Financial support and sponsorship
None.
Conflict of interest
None.
Ethical approval and consent to participate
Approved by Institutional Ethical Committee Dr. Sampurnanand Medical College Jodhpur. Certificate reference number: SNMC/IEC/2021/plan/387.
Consent for publication
Not applicable.
References
1. AJ, M., TC, H., ML, P., LC, S., & WR, J. (1999). Guideline
for Prevention of Surgical Site Infection, 1999.
Centres for Disease Control and Prevention (CDC)
Hospital Infection Control Practices Advisory Committee.
American Journal of Infection Control, 27,
97–132.
2. Owens, C. D., & Stoessel, K. (2008). Surgical site
infections: epidemiology, microbiology and prevention.
Journal of Hospital Infection, 70 (Suppl 2),
3-10.
3. Weigelt, J. A., Lipsky, B. A., Tabak, Y. P., Derby, K. G.,
Kim, M., & Gupta, V. (2010). Surgical site infections:
Causative pathogens and associated outcomes.
American Journal of Infection Control, 38(2), 112-
120.
4. Ezechi, O. C., Fasubaa, O. B., & Dare, F. O. (2000).
Socioeconomic barriers to safe motherhood among booked patients in rural Nigerian communities.
Journal of Obstetrics and Gynaecology Research,
20(1), 32-34.
5. Chaim, W., Bashiri, A., Bar-David, J., Shoham-Vardi,
I., & Mazor, M. (2000). Prevalence and clinical signi
ficance of postpartum endometritis and wound
infection. Infectious Diseases in Obstetrics and Gynecology,
8(2), 77-82.
6. Killian, C. A., Graffunder, E. M., Vinciguerra, T. J., &
Venezia, R. A. (2001). Risk factors for surgical-site
infections following cesarean section. Infection
Control and Hospital Epidemiology, 22(10), 613-
617.
7. Johnson, A., Young, D., & Reilly, J. (2006). Caesarean
section surgical site infection surveillance. Journal
of Hospital Infection, 64(1), 30-35.
8. Webster, J. (1988). Post-caesarean wound infection:
a review of the risk factors. Australian and
New Zealand Journal of Obstetrics and Gynaecology,
28(3), 201-207.
9. Mitt, P., Lang, K., Peri, A., & Maimets, M. (2005).
Surgical-site infections following cesarean section
in an Estonian university hospital: postdischarge
surveillance and analysis of risk factors. Infection
Control and Hospital Epidemiology, 26(5), 449-
454.
10. Litta, P., Vita, P., Konshi, d. T., J., & Omnis, G. (1995).
Risk factors for complicating infections after Caesarean
section. Clinical and Experimental Obstetrics
& Gynecology, 22, 71-75.
11. Beattie, P. G., Rings, T. R., Hunter, M. F., & Lake, Y.
(1994). Risk factors for wound infection following
caesarean section. Australian and New Zealand
Journal of Obstetrics and Gynaecology, 34(4), 398-
402.
12. Gorbach, S., Bartlett, J., & Blacklow, N. (2004). Surgical
Site Infections. Infectious Diseases, 3rd edition,
823-829.
13. Lilani, S., Jangale, N., & Chowdhury, A. (2005). Surgical
Site Infections in clean and clean-contaminated
cases. Indian Journal of Medical Microbiology
23, 249-252.
14. Prevention, C. f. D. C. a. (2017). Surgical site infection
(SSI) event: procedure-associated module.
15. (CLSI), C. a. L. S. I. (2006). Performance standards
for antimicrobial susceptibility testing. 16th informational
supplements, CLSI Document M2-A9.
16. Negi, V., Pal, S., Juyal, D., Sharma, M. K., & Sharma,
N. (2015). Bacteriological Profile of Surgical Site
Infections and Their Antibiogram: A Study From
Resource Constrained Rural Setting of Uttarakhand
State, India. Journal of clinical and diagnostic research,
9(10), Dc17-20.
17. Cunningham, F. G., Levero, K. J., & Bloom, S. L.
(2018). Williams Obstetrics. 25th Edition, McGraw-
Hill, New York, 819-822.
18. Njoku, C. O., & Njoku, A. N. (2019). Microbiological
Pattern of Surgical Site Infection Following Caesarean
Section at the University of Calabar Teaching
Hospital. Open Access Macedonian Journal of
Medical Sciences, 7(9), 1430-1435.
19. Ward, V. P., Charlett, A., Fagan, J., & Crawshaw, S. C.
(2008). Enhanced surgical site infection surveillance
following caesarean section: experience of a
multicentre collaborative post-discharge system.
Journal of Hospital Infection, 70(2), 166-173.
20. Wendmagegn, T. A., Abera, G. B., Tsehaye, W. T.,
Gebresslasie,
K. B., & Tella, B. G. (2018). Magnitude
and determinants of surgical site infecion among
women underwent cesarean section in Ayder
comprehensive specialized hospital Mekelle City,
Tigray region, Northern Ethiopia, 2016. Bmc Pregnancy
and Childbirth, 18(1), 489.
21. G., G., & H, Z. (2016). Incidence of Post Cesarean
Section Wound Infection and its Associated Risk
Factors in Orrota National Referral Maternity Hospital.
International Journal of Allied Medical Sciences
and Clinical Research, 4(3).
22. Shapiro, M., Muñoz, A., Tager, I. B., Schoenbaum, S.
C., & Polk, B. F. (1982). Risk factors for infection at
the operative site after abdominal or vaginal hysterectomy.
the New England Journal of Medicine,
307(27), 1661-1666.
23. Tran, T. S., Jamulitrat, S., Chongsuvivatwong, V., &
Geater, A. (2000). Risk factors for postcesarean
surgical site infection. Obstetrics and Gynecology,
95(3), 367-371.