Open Access | Research Article
This work is licensed under a
Creative Commons Attribution-ShareAlike 4.0 International License.
Management of acute sigmoid volvulus in a tertiary hospital north central nigeria: The place of advanced radiodiagnostics
* Correspondence to: Edwin Oseni-Momodu
Mailing address:
Department of Surgery, College of Medicine and Health Sciences, Jos
campus, Bingham University Teaching Hospital, Bingham University, Zaria
Bypass, PMB 2238, City Postcode 930241, Jos. Plateau State. Nigeria.
E-mail:
eosenimomodu@yahoo.com
Received: 09 March 2021 / Accepted: 02 August 2021
DOI:
10.3149/CSRC.2021.09.079
Abstract
Background: Volvulus involves the twisting of an air-liquid
stool-filled thin-walled segment of an intestine around its
necessarily narrow mesenterial base, thereby strangulating the blood
vessels, which often causes necrosis of this redundant intestinal
segment. Intestinal volvulus had always been supposed to be a disease
of the blacks from West African and the Bushmen natives of South
Africa. The West African subset became the index region. Surgery was
the best treatment for the full-blown disease. Conservative methods of
management have only just been developed and studied in the
Scandinavian countries. Scandinavian early rectosigmoidoscopic
reductions of the twisted colonic segment have offered some valuable
alternative helps.
Methods: We studied the documents of all the 44 patients who
had a presumptive diagnosis of acute or subsubacute colonic volvulus
and were admitted to surgical management. Biostatistics, exact history
taking of the patients, carefully structured physical examination, and
a good digital scout X-ray investigation of the abdomen helped to make
a rapid diagnosis. Laparotomies confirmed such a diagnosis. We did not
regularly attempt to do recto-sigmoidoscopic untwisting of the
volvuli. All had Hartmann’s procedure surgeries with terminal
colostomies.
Results: Of the 41 patients admitted to surgical management, 31
were males and 10 were females with a ratio of 3:1. The timing of
surgery influenced mortalities and morbidities greatly.
Conclusion: The diagnosis of acute volvulus was simple. We
needed to record the medical history, took the proper physical
examination, correctly explained the examination results, and only
studied the abdominal Xray film without resorting to advanced computer
topography
Keywords
Absolute wind stoppage, safety checklist, dolichol-sigmoid, endemicity, Hartmann’s procedure, primary anastomosis
Introduction
Volvulus occurs when an air-filled thin-walled segment of the intestine
twists about its narrow mesentery [1]. Colonic volvulus
occurs up to 90% at the colo-sigmoid part and 20% at the caecum
[1]. Any redundant portion of the colon can be affected by
volvulus. Most patients are males [2-7]. A volvulus may
reduce spontaneously but more commonly causes bowel obstruction, which
can progress to strangulation, gangrene, and perforation. Chronic
constipation may lead to a large, redundant colon that predisposes to
volvulus, especially if the mesenteric base is narrow [3-6].
Anatomical studies suggest that Black-Africans have a predisposition to
sigmoid volvulus, with a male gender bias due to an elongated narrow
mesenterial base of the sigmoid colon segment [1, 2].
Previous studies have shown that the elongated sigmoid colon in Black
Africans presents in utero more frequently in Black-African males
[2, 3]. For instance, a narrow-shaped mesentery and a
dolichol-sigmoid segment, with increased splanchnic pressure, commonly
leads to necrosis and perforation in male fetuses, while broadly shaped
sigmoid occurs more in females [6]. This anatomy explains the
preponderance of volvulus amongst males when compared to females
[6-8]. Delayed patients’ referral and patients’
health-seeking behaviors contribute to necrosis of the volvulus segments
and their twisted knots and increase splanchnic pressure, which causes
the perforations [6].
The non-resection procedures of
sigmoidopexy and recto-sigmoidoplasty do not require bowel preparation
with lower morbidity and mortality rates at first incidence. However,
the recurrence rate is still very high, ranging from 45% to 71%
[9].
Where a detorsion fails, and there are signs of colonic
gangrene, sigmoid resection and Hartmanns’ procedure with a temporary
colostomy with a mucus spout would be done to avoid the high mortality
associated with primary anastomosis in this situation
[6, 10].
An 180° twisting of the rectosigmoid, while
considered physiological and reduces spontaneously in 2%, torsion of
more than 180° causes ischemia and necrosis. The preferential
anticlockwise direction of twisting in circa 70% as reported in the
literature remains still unexplained [6, 11].
Materials and methods
Patients
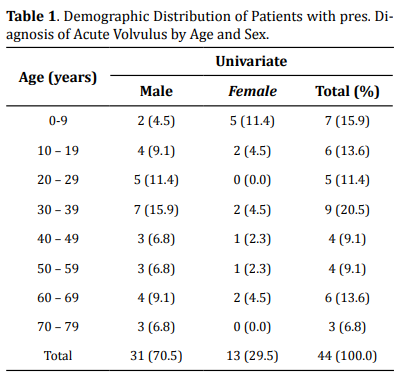
The documents of the forty-four patients that were admitted through the A and E were initially taken into this study. The basic information of all the patients is presented in Table 1. Three patients were false-positive at laparotomies, in that they did not have sigmoid volvulus: they were excluded from this study.
Surgeon
Lead surgeons were consultant general surgeons of the same level with a subspecialty in visceral surgery. Other surgeons were either Fellows of the International College of Surgeons (FICS), the National Postgraduate Medical College of Nigeria, or Surgeons of the West African College of Surgeons (FWACS) and foreign surgeons of equivalent standing. They were ably assisted by in-house Senior Registrars honing their gastrointestinal surgery experiences and were either first or second assistants.
Management procedures
All patients had generalized abdominal pain and tenderness of left upper abdominal quadrants, vomiting, and gross abdominal distensions; constipation, and absolute wind stoppage were also present [5]. These usual features of acute sigmoid volvulus were not difficult to recognize, especially when gross abdominal gaseous distention of the upper left abdominal quadrant with an emptiness of the left iliac depression were present [6, 12].
Imaging
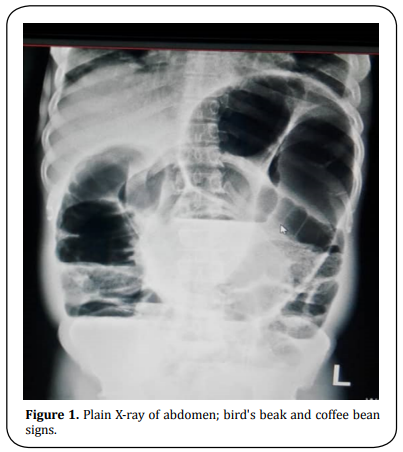
We did not need to do a CT scan nor MRI. Volvulus diagnosis was already clinched at the surgical outpatient because of these specific signs and symptoms, especially various peculiar radiological findings of the plain abdomen such as birds’ beak, coffee bean signs that were confirmed at laparotomy (Figure 1).
For the first 45 minutes of the vigorous
resuscitation, 1 liter of crystalloid fluid was administered followed by
1 liter of crystalloid fluid 6 hours till the operation was done. All
patients who consented to the operation and were willing to participate
in the retrospective study were given informed consent. They explained
the need for a colostomy, which can be reversed after three to six
months at home. Those who agreed were also included in the study.
Twenty-four laparotomies were performed as emergencies on the same
nights of admission because the American Society of Anesthesiologists’
(ASA) Physical Status Classification System for these patients was
already about III. Sixteen patients were operated on the following
mornings who had ASA Physical Status Classification System much less
than III. The average time of resuscitation and preparation for surgery
was 2-3 hours.
Inclusion criteria
1、Initial inclusion criteria
• All patients admitted for management as surgical inpatients who
consented to the study by signing their informed consent forms.
• All following selections were done as eligible patients gave consent
to the study.
• Patients’ names were pass-worded thereby guarantying their
anonymity.
• All patients were assured anonymity and the freedom to quit the study
at any time if they so wished.
• Guardians of minors signed for their wards after the surgeons offered
sufficient explanations.
2、Novel secondary inclusion criteria developed by us in-house
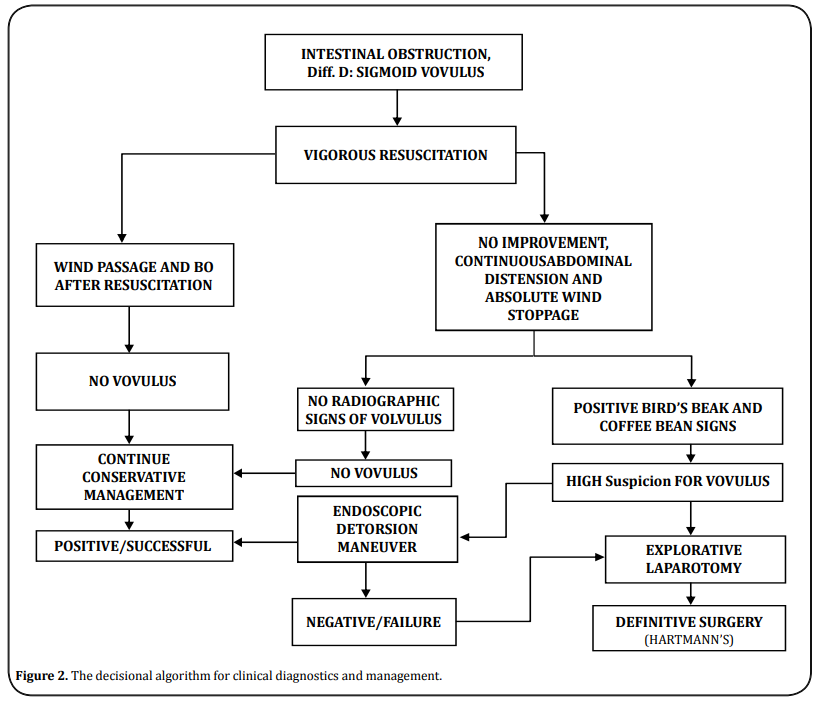
Our in-house developed surgical protocol, a decisional algorithm of clinical or radiologic severity, was used to further select the patients. The details were shown in Figure 2.
Patients were bladder
catheterized and given preoperative broad-spectrum antibiotics. They
also received intra-venous cephalosporin, aminoglycosides, and infusions
of metronidazole before and during surgeries, when lasted beyond 3
hours. Blood specimens were collected, and 2-3 pints of whole blood were
grouped and crossed matched before taking the patients to the OR.
Infusion of one pint of whole blood was begun at the start of surgery,
even if PCV was normal.
The operating team regularly carried out
operation room (OR) debriefings and safety checklists for patients. The
checklists included: (1) Sign-in consisting of a complete check of
patients’ details before induction of anesthesia. The patient’s
identity, the procedure, the side/site of the body, consent form, and
the pulse oximeters’ function were cross-checked; (2) Time-out, which
takes place before skin incision when surgeons and entire team identify
themselves by names were read out loud by the lead surgeon; (3)
Sign-out; undertaken by operation room (OR) nurse, who calls out the
entire team. Sign-out was done after skin closure. Sponges, instruments,
swabs, were counted at this time, etc. Each part of these debriefings
was implemented at defined time-points by the team working together
under each named persons’ responsibility.
Laparotomies were done
through midline incisions beginning 2-4 cm above the navel and driven
left round it down to 2-4 cm above the pubic bone. Deepening into the
abdominal cavity was mainly non-eventful, care being taken not to
perforate the massively distended colonic segments. Inspection and
palpations were carefully done to both identify the twisted knots and
rule out necrosis and perforations.
If the remaining proximal piece
of the descending colon was long enough, a colostomy position was chosen
at a site below the expected beltline; otherwise, it was chosen higher.
A colostomy with a spout was now fashioned such that a brook would not
develop, which process required proficiency. The stoma was sutured to
the skin with absorbable suture material, Vicryl-0 ®. A well-fashioned
colostomy began to function in tabula. We recorded 5 instances of stoma
prolapses and anastomotic complications.
Anastomotic complications
were diagnosed and managed along the lines of management of
entero-cutaneous fistulas with typical clinical presentations shortly
after surgery within few days. Lead symptoms were:
• Fever;
• Erythematous wound(s);
• When a few skin sutures were removed, purulent or bloody discharge was
noted;
• Leakage of enteric contents then occurred, sometimes immediately, but
often within 1 or 2 days after stitch removal.
When the diagnosis
was in doubt, confirmation was achieved by injection of a water-soluble
contrast medium into the fistula, such as gastrografin or conray® or
barium. In this way, the presence and extent of any peritoneal abscess
cavities were be delineated.
Further management included the
classification of the resulting fistulas by location and volume of daily
fistula output. Most daily outputs were less than 200ml of stool mixed
with serosanguinous secretions. Specialized treatment required the
establishment of controlled drainage, using a sump suction apparatus.
Management of sepsis, prevention of fluid and electrolyte depletion, and
skin protection were ensured. Provision of adequate nutrition was made
locally by oral feeding with protein-enhanced corn porridge, consisting
of groundfish, and eggs. The control of fistula output was most easily
accomplished by intubation of the fistula tract with a drain and
suctioning when the need arose.
Statistical data analysis
The statistical analysis was performed using Excel and Statistical Package for the Social Sciences (SPSS) 22nd Edition.
Results
Inclusion of patient information and treatment costs
The average age of all patients was 65 years with a median age of 38 years, median sex distribution of 1.00, and a standard deviation of 0.454. The median length of hospital stay (LOS) was 7.0 days, and a standard deviation of 24.5 days. The average hospital treatment cost for a patient with a LOS of 7.0 days was N170, 000 ($454.2), at the current bank exchange rate. There were hardly any intra-operative complications since the surgery was straightforward as preplanned.
Treatment modalities
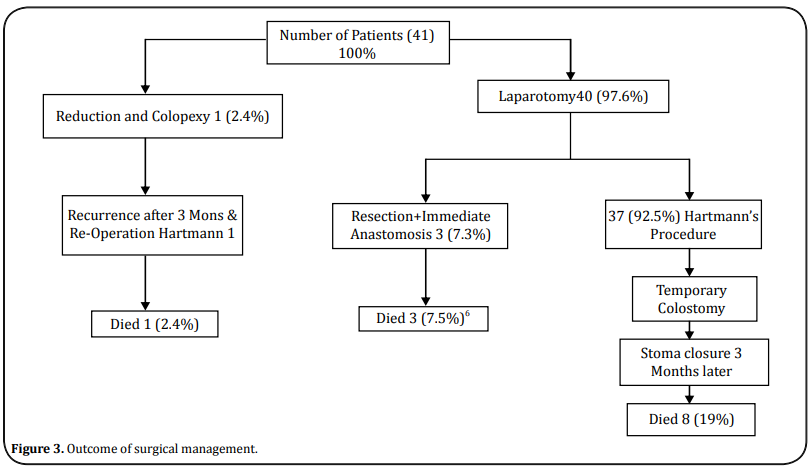
Forty-one patients (100%) made our inclusion criteria for this study and underwent explorative laparotomies. Thirty-seven patients (90.2%) had amputations of the necrotic colon segments and Hartmanns’ procedures. Colostomies with spouts were formed at convenient points of the left upper abdominal quadrant. The sigmoid-rectal stumps were closed with distinct non-absorbable sutures for easy identification in the event of reversal surgeries later. One male patient (2.4%) had a reduction and colopexy since there was no necrosis. Three males (7.3%) had resection of the necrotic segment and an immediate end-to-end anastomosis with on-table minimal lavage of the rectal stump per rectum (Figure 3).
Twisted mesenterial knots were identified in all patients; twenty-three
patients had frank necrotic segments with varied sizes and numbers of
perforations and peritoneal spillage of stool. Severe abdominal spillage
of stool from perforations of necrosis of the twisted colon segment
negatively affected morbidity and mortality values [1, 6].
The procedure involved removing the enlarged necrotic colon segments
after first clamping about 2-3 cm into its healthy proximal colon with 2
non-crushing intestinal clamps; this procedure was repeated 2-3 cm
distal from the necrotic end. The segment was resected between the
applied intestinal clamps, carefully unwound, being held between
abdominal packs, and lifted out of the abdomen, care was taken not to
allow more leakage into the peritoneal cavity in the hope of reduction
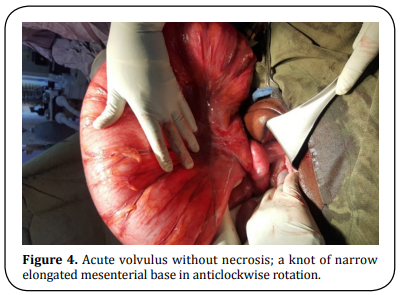
of post-operative infection (Figure 4-5).
In Figure 4, the results
showed one of our acute volvuli without necrosis; the volvulus segment
is elongated with a narrow mesenterial base, which is the essential
contributory factor for the twist; in this case, the torsion is greater
than 180° [3-5].
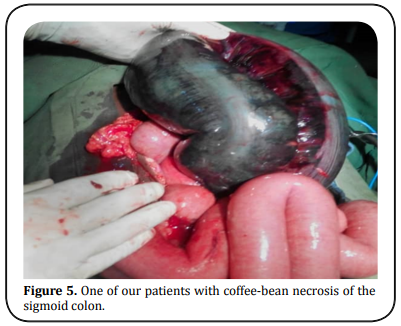
Treatment outcome
Our mortality cases included the death of the one patient (2.4%) that
had a reduction and colopexy but a recurrence of volvulus three months
later; he died after the subsequent Hartmanns’ procedure. Thirty-eight
(90%) patients had Hartmanns’ procedures. Fifteen (48%) colostomies
reverted after three months of recuperation at home were alive at the
end of this study.Others were lost to the study.
One male patient
had a reduction of volvulus and colopexy without relief colostomy and
on-table colonic lavage. The surgeons did an early per rectum
post-operative wash out of the rectum to enhance the early onset of
bowel movements. We had one recrudescence of the volvulus about six
months later [6].
Forty Patients (97.6%) in the male to
female ratio of 27:13 underwent the Hartmanns’ procedure. The mean
operation time was 3 hours. The median duration of the procedures was
180 minutes. The time lag between admission and surgery was a median of
8 hours.
The immediate survival rate of the operated cases was
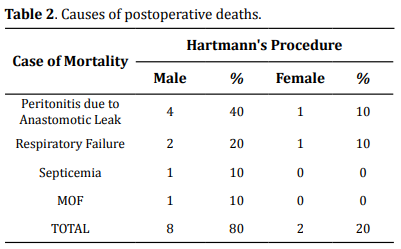
twenty-eight (70%). The mortality rate was twelve (30%) from various
post-operative morbidities, especially peritonitis, septicemia,
respiratory failure; Table 2. The rest died of consequences of
multi-organ failures (MOF) [9].
Discussion
We had a high mortality rate of 19% for Hartmanns’ procedure alone. This
2-digit percentage, which is comparable to the mortality rate of 16.7%
in Kaduna, Nigeria found by Udueze et al. [13] and is higher
than rates of 10.5% in the Gambia [1] and 12% at the
University Hospital of Cocody, Abidjan [11] and 12% in South
Africa [14].
The young stage of development of the BHUTH
being five years of existence could explain this high mortality rate at
the commencement of this study.
Our three cases who had immediate
continuity anastomosis after resection of volvulus died; they posted
high mortality of 7%, which was similar to the previously reported
conclusion [14]. This 100% (3 cases) of mortality for our
immediate, primary continuity anastomosis was discouraging. We could not
continue this type of management of acute volvulus until the required
operations skills had been honed. This mortality rate is in
contradistinction to the results of one-stage emergency resection and
primary anastomosis for sigmoid volvulus by Naseer et al.
[15] and Sule et al. [16] in their one-stage
procedure in the management of acute sigmoid volvulus without colonic
lavage. The smallness of our patients’ population for this type of
surgery would have mitigated our results. A prospective study of a
larger patients’ subset would help better the learning curve
eventually.
The average occurrence per year of 9% sigmoid volvulus
in our study appears low, despite being in the endemic sub-region
compared to similar studies [17]. Acute volvulus occurred
highest in the age group of 30-39 years with 21% and male to female
ratio of 7:2 in keeping with higher male prevalence as shown in all the
age subset [6] (Table 1).
The male to female ratio of 31
to 13, was less than that of Kassi et al. [11]. Our patients’
mean age of 65 years with a median of 38 years tallied with the
previously reported studies [1, 11]. The male to female ratio
of volvulus in the age group of 0-19 years was 7:4. The male
preponderance is in keeping with trends of the works of Perrot et al.
[6], and others in the endemic areas by the previously
reported researches [11, 17].
All cases of sigmoid
volvulus in this study occurred just above the iliac crest, where the
descending colon becomes sigmoid colon without sex difference in
conformity with the works of Mnguni et al. [9].
Most
patients were in a good state of nourishment despite a history of
chronic recurrences of constipations. Their vital signs were not
alarming, nor were their work-up findings deranged
[6].
Excision and primary continuity anastomosis
[2, 18] were done when there was no intestinal necrosis and
the remaining colons’ length sufficient for the anastomosis without
tension [6]. The high rate of recurrence of volvulus by
successful endoscopic detorsion in literature at about 60% within a
short time did not impart a drive to pursue the procedure
[5, 6].
Our mortality rate of twelve (29%) was higher
than the 10.5% found in the Gambia [1] and was higher than
the 12% at the University Hospital of Cocody, Abidjan
[17].
When combined, peritonitis and septicemia of 50%
(Table 2) due to anastomotic leak accounted for the significant
mortality of 29 % in our study, as shown in the literature
[9, 12]. However, the ease of restoration of intestinal
continuity when the availability of a good length of rectal stump made
Hartmanns’ procedure the gold standard despite the temporary
post-operative disabilities and immediate worsening of quality of life
(QOL).
Our experience with primary anastomosis following resection
of the gangrenous bowel segment was not satisfactory as would be in more
experienced hands. We created an end colostomy with a cuff and left the
closed-up distal sigmoid-rectum in situ. They were always too short for
reversal surgery.
Subordinate surgeons were not encouraged to do Hartmanns’ procedure
alone. In the absence of sufficient length of rectal stump, reversal of
Hartmanns’ procedure was not done [5, 11]
Conclusion
Our study suggested that the management of acute sigmoid volvulus with
excision of the necrotic segment and creating a continuity primary
anastomosis in the same sitting was fraught with high mortality as a
result of post-operative anastomotic breakdown [6]. Thus,
Hartmann’s procedures should be performed when possible, especially when
the sigmoid segment is gangrenous and the rest rectum stump short.
We
hope that this paper will encourage general surgeons in regional
hospitals to perform more Hartmann’s procedures, rather than referring
patients a priori to tertiary centers which are far away just because of
the unavailability of CT-scan and MRI.
Declarations
Acknowledgments
Statistical reviews were done by Mr. Henry Job of the ICT Department of BHUTH.
Authors’ contributions
Oseni-Momodu, EO: Conception, design, drafting and writing and revisions of the manuscript, Shehu, H: Administrative and critical revisions of the manuscript, Lee, A J: Lead Surgeon in most cases and literature search and photography There was no financial support nor sponsorship. All authors agreed to pay for the cost of publication if the need arises.
Conflict of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Approval to undertake this study was applied for and received from the Human Research and Ethics Committee of Bingham University Teaching Hospital (BHUTH). Approval number: NHREC/21/05/2005/00710.
Consent for publication
All authors consented to publish this paper.
References
1. Nuhu, A., & Jah, A. (2010). Acute sigmoid volvulus in a West African
population. Annals of African Medicine, 9(2), 86-90.
2. Madiba, T. E., Aldous, C., & Haffajee, M. R. (2015). The morphology
of the foetal sigmoid colon in the African population: a possible
predisposition to sigmoid volvulus. Colorectal Disease, 17(12),
1114-1120.
3. Madiba, T. E., & Haffajee, M. R. (2011). Sigmoid colon morphology in
the population groups of Durban, South Africa, with special reference to
sigmoid volvulus. Clinical Anatomy, 24(4), 441-453.
4. Madiba, T. E., & Haffajee, M. R. (2010). Anatomical variations in the
level of origin of the sigmoid colon from the descending colon and the
attachment of the sigmoid mesocolon. Clinical Anatomy, 23(2),
179-185.
5. van der Naald, N., Prins, M. I., Otten, K., Kumwenda, D., &
Bleichrodt, R. P. (2018). Novel Approach to Treat Uncomplicated Sigmoid
Volvulus Combining Minimally Invasive Surgery with Enhanced Recovery, in
a Rural Hospital in Zambia. World Journal of Surgery, 42(6),
1590-1596.
6. Perrot, L., Fohlen, A., Alves, A., & Lubrano, J. (2016). Management
of the colonic volvulus in 2016. Journal of Visceral Surgery, 153(3),
183-192.
7. Maddah, G., Kazemzadeh, G. H., Abdollahi, A., Bahar, M. M.,
Tavassoli, A., & Shabahang, H. (2014). Management of sigmoid volvulus:
options and prognosis. Journal of the College of Physicians and Surgeons
Pakistan, 24(1), 13-17.
8. Samuel, J. C., Akinkuotu, A., Msiska, N., Cairns, B. A., Muyco, A.
P., & Charles, A. G. (2010). Re-examining treatment strategies for
sigmoid volvulus: An analysis of treatment and outcomes in Lilongwe,
Malawi. Global journal of Surgery, 1(2), 149-153.
9. Mnguni, M. N., Islam, J., Manzini, V., Govindasamy, V., Zulu, B. M.,
Clarke, D. L., & Madiba, T. E. (2012). How far has the pendulum swung in
the surgical management of sigmoid volvulus? Experience from the
KwaZulu-Natal Teaching Hospitals and review of the literature.
Colorectal Disease, 14(12), 1531-1537.
10. Ghariani, B., Houissa, H., & Sebai, F. (2010). [Management of
sigmoid volvulus]. Tunisia Medical, 88(3), 163-167.
11. Kassi, A. B., Lebeau, R., Yenon, K. S., Katche, E., Diane, B., &
Kouassi, J. C. (2011). Morbidity and mortality of Hartmann’s procedure
for sigmoid volvulus at the University Hospital of Cocody, Abidjan. West
African Journal of Medicine, 30(3), 169-172.
12. Atamanalp, S. S. (2013). Treatment of sigmoid volvulus: a
single-center experience of 952 patients over 46.5 years. Techniques in
Coloproctology, 17(5), 561-569.
13. Udezue, N. O. (1990). Sigmoid volvulus in Kaduna, Nigeria. Diseases
of the Colon & Rectum, 33(8), 647-649.
14. Mokoena, T. R., & Madiba, T. E. (1995). Sigmoid volvulus among
Africans in Durban. Tropical & Geographical Medicine, 47(5), 216-217.
15. Naseer, A., Ahmad, S., Naeem, M., & Safirullah. (2010). One stage
emergency resection and primary anastomosis for sigmoid volvulus.
Journal of the College of Physicians and Surgeons Pakistan, 20(5),
307-309.
16. Sule, A. Z., Misauno, M., Opaluwa, A. S., Ojo, E., & Obekpa, P. O.
(2007). One stage procedure in the management of acute sigmoid volvulus
without colonic lavage. Surgeon, 5(5), 268-270.
17. Mulas, C., Bruna, M., García-Armengol, J., & Roig, J. V. (2010).
Management of colonic volvulus. Experience in 75 patients. Revista
Espanola De Enfermedades Digestivas, 102(4), 239-248.
18. Liang, J. T., Lai, H. S., & Lee, P. H. (2006). Elective
laparoscopically assisted sigmoidectomy for the sigmoid volvulus.
Surgical Endoscopy And Other Interventional Techniques, 20(11),
1772-1773.